Worm Breeder's Gazette 12(2): 77 (January 1, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
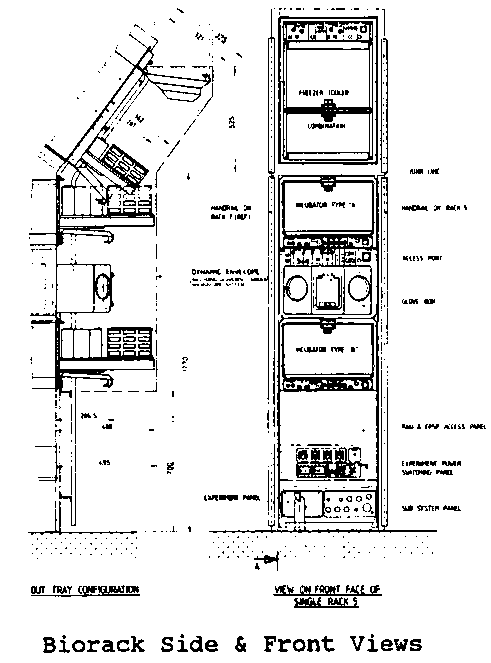
On January 22, 1992 C. elegans will take its first trip into space aboard the space shuttle Discovery as part of the International Microgravity Laboratory #1 mission ( STS-42 ).A crew of seven will operate the Spacelab which contains a variety of physics and biology experiment facilities mounted in "racks" along its walls. One of these, the Biorack, is a European Space Agency device that is essentially a miniaturized biology laboratory with a microscope, glove box, incubators, freezers and a series of centrifuges that provide 1 X Gravity acceleration controls for individual experiments. Each experiment package is designed to fit into a series of small aluminum boxes (Biorack Type I or Type II containers) which interface with the Biorack's equipment. 17 investigations from 15 laboratories (12 in Europe & 3 in the U. S.) will share the Biorack facility during the 7 day mission. We will use 13 of the Biorack's containers and have designed our experiment to operate passively once loaded on the ground at Cape Canaveral.
Our experiment is designed to investigate the effects of cosmic rays with respect to mutation and the effects of gravity with respect to development and meiosis. The cosmic ray issue is of increasing concern to NASA as longer and longer space flights are being planned and there are unique effects of charged particle radiation which contribute to health risks.
[See Figure 1]
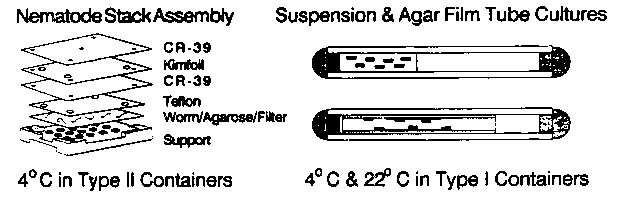
The development and meiosis issues center around the potential role of gravity in establishing early embryonic asymmetry or in orienting centrioles and chromosomes. The first objective is to verify that charged particles in space induce genetic damage with the same effectiveness as observed on the ground where monoenergetic beams of particles from accelerators are used to simulate this radiation. The dose, dose rate and mixture of radiation species and energies observed in space cannot be accurately reproduced on the ground so that a model of expected results is constructed from interpolations and extrapolations of accelerator experiment results. A second variable which may contribute to the overall response is the absence of gravity which can directly or indirectly alter the physiology of animals and even single celled organisms. It is possible that microgravity will perturb the repair of cosmic ray-induced damage in worms. To detect genetic damage we will employ the eT1 (III:V)balancer technique developed by Rosenbluth et al. to isolate lethals on LGV and LGIII in a 350-400 gene region. To supplement this measurement we will isolate unc-22 mutants in N2 which will be used for hybridization studies of mutant structure rather than for kinetics. In order to minimize handling we will use dauer larvae as the target animals. The low dose of radiation expected on this mission (<1rad) requires a large sample size and we expect to use about 10+E5 petri dishes for the mutant hunts. During the flight one group of dauers will be suspended in M9 buffer in lexan tubes with fixed air and liquid volumes and placed at several locations within the Spacelab and Biorack so as to vary radiation shielding values. Another group will be immobilized on nitrocellulose filters under 4% agarose made up with modified S medium and held at 4°C. These filters are assembled into a laminated "sandwich" with plastic nuclear track detectors developed by the USF team. These plastics can be treated with strong alkali to etch away plastic damaged by cosmic ray particles. The resulting "etch cone" morphology correlates with particle charge and energy loss rate and the trajectories identify worms struck by, or in the immediate vicinity of, the cosmic rays. The latter configuration allows us to correlate individual cosmic rays with worms in apposed layers.
[See Figure 2]
In order to investigate whether chromosome mechanics are normal at meiosis and whether development is successfully completed under microgravity we will inoculate tubes with three different genotypes of animals. The tubes are lined with a thin cylindrical agar film and inoculated with E. coli K12 X1666 controlled with kanamycin and streptomycin. (Due to confinement of worms and bacterial food within closed tubes the bacteria could not be allowed to actively metabolize and rob worms of oxygen.) The final conditions are roughly equivalent to a sealed 35mm mating plate with cylindrical geometry. One experiment places dpy-11 dauer larvae and young adult him-5 males together in individual tube cultures. Due to the shuttle launch and operational schedules the hermaphrodites will complete most of gametogenesis and become competent for mating only after the shuttle is in orbit and "microgravity" conditions (10+E-5 X gravity) are established . If mating is successful and if transmission ratios are normal then half of WT outcross progeny will be males. All F1 progeny will have been conceived under microgravity or at 1XG on control centrifuges. Following shuttle landing, F1 'swill be scored for phenotypic ratios, then by immunofluorescence for distribution of cytoplasmic determinants in embryos and cell nuclei distribution in all stages. The second and third experiments test the Mendelian segregation ratios for linked and unlinked marker mutations and also serve as sources of animals for immunofluorescence examinations. Prior to launch him-5 males will be mated to dpy-17 unc-32 III and dpy-18 III: unc-17 IV hermaphrodites. F1 L4 heterozygous hermaphrodites will be loaded into tubes and sealed. During the flight animals should be able to complete up to two rounds of reproduction. If chromosome mechanics are unperturbed by microgravity the F2 'sshould exhibit the usual 3:1 (+ recombinants) or 9:3:3:1 phenotypic ratios. Some F2 'swill be homozygous for him-5 and will go on to segregate F3 males of all phenotypes. The presence of marked males will be genetic proof of two successful rounds of reproduction. Why suspect microgravity effects? Here are some examples of observations from spaceflight experiments. To date, no multicellular organism has successfully completed a continuous full life cycle under microgravity conditions although all segments of life cycles have been successfully completed in organisms such as Drosophila and Arabidopsis. In chicken embryos fertilized at different times before launch, embryogenesis in microgravity is arrested in a stage specific fashion. Lectin stimulated mitogenesis in mouse and human lymphocytes is almost completely inhibited in microgravity and is proportional to gravity levels above 1XG. In the stick insect, Carausius, embryos struck by cosmic rays show a stage specific synergism with microgravity for embryonic anomalies. Catastrophic levels of chromosome breakage and other anomalies have been observed in plant meristem cells from several spaceflight experiments with very low radiation exposures. These and other observations indicate that direct or indirect (eg. altered mass transport due to absence of convection) influences of gravity unloading may significantly perturb development and physiology, perhaps in worms as well.