Worm Breeder's Gazette 12(2): 72 (January 1, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
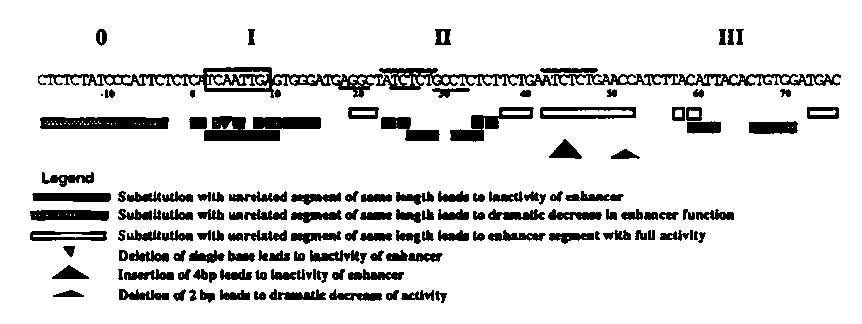
The unc-54 gene contains a body wall specific enhancer element located in the third intron (WBG 11#2 p. 2). This element is defined by its ability to induce body wall expression of ß-Gal when placed upstream of a [pharyngeal muscle specific] myo-2 ::lacZfusion. Using this assay, we narrowed the functional enhancer sequence down to a 90 base pair fragment (which we will refer to as the '90-mer').
As the myo-2 promoter is already muscle specific, we also tried to test this enhancer element in the context of a more promiscuous promoter. Placing the 90-mer upstream of a glp-1 promoter-lacZ fusion construct results in detectable body wall muscle staining (although the level and frequency of body wall activation is much lower than when the myo-2 promoter was used). When a metallothionein or minimal heat shock promoter was used no activation of expression in body wall muscle was seen (thanks to J. Freedman & C. Rubin for the MT gene and G. Seydoux for the HSP construct). Although this enhancer element is capable of activating "non muscle" promoters to express in muscle it certainly shows a preference for muscle promoters.
Site directed mutagenesis has been used to determine which sequences within the 90-mer are required for enhancer function (as assayed initially by ability to activate the myo-2 promoter in body wall muscle). As summarized in the figure, the 90-mer contains four essential sites. Separating these sites are generally regions that can be replaced without eliminating enhancer function. In at least one case (the region between site II and site III) the intervening region plays a spacing role: although this region can be substituted with unrelated sequences of the same length, small deletions or insertions in the region lead to a loss or a dramatic decrease in enhancer activity. Considering the complexity of this enhancer element it seems very likely that its activity is regulated by multiple factors.
We also tested the multimerized subelements upstream of glp-1 fusions. Multimerized sites 0 and I activate weak body wall and vulval muscle expression from the glp-1 promoter segment. Multimerized site II does not show any activation of the glp segment, while multimerized site III activates the glp-1 promoter construct strongly, generating primarily expression in hypodermal cells. As the individual subelements activate the glp-1 promoter in different tissues we consider the possibility that this enhancer element might function in response to both muscle specific and more general factors.
In screens of a cDNA expression library (see previous abstract for a description of the library) a set of cDNAs encoding a DNA binding protein of the 'Y Box' class have been isolated. This protein has strong affinity for sites 0 and I and we are currently investigating the possibility that it plays a central role in activation of the enhancer.
[See Figure 1]