Worm Breeder's Gazette 12(1): 41 (September 1, 1991)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
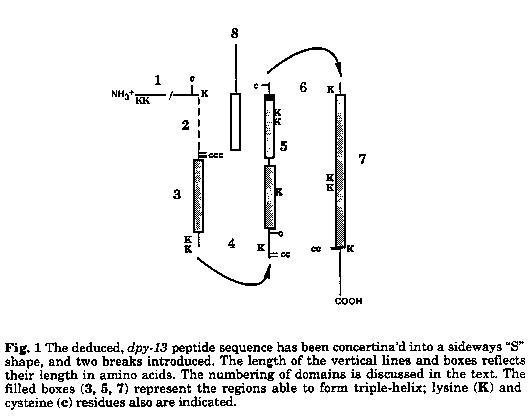
Using a combination of computer and hand alignments, we have generated a model for the dpy-13 product in which the peptide folds back on itself to form a triple helical structure with non-helical termini (telopeptides) and a region of potential interaction with other collagens. This model is consistent with a proposal based on a classical, physical biochemical analysis of Ascaris collagen (1) (McBride and Harrington, Biochem., 6: 1484, 1967). Key features of our model are:
1) The four stretches of sequence able to form triple-helix define three domains (3, 5 and 7 in Fig. 1). Domains 5 and 7 are available to participate in helix formation along most of their length, but domain 3 is sufficient to interact with only the amino-half of 5 and the carboxy half of 7. Perhaps the hypervariable domain (2) interacts with the remainder of domains 5 and 7. Alternatively, another peptide (8) with a gly-X-Y motif might contribute to triple-helix. The discontinuity in 5 possibly is required for such interactions.
This folding arrangement places the second gly-X-Y region (5) in anti-parallel to the other two (3 and 7), effectively reversing X and Y order. This reversal enhances the preferential distribution of the bulky residues in the X position (Traub, Israel J. Chem., 12: 435, 1974) and places six of the seven proline residues in suitable locations for X/Y pairing (Jones and Miller, JMB, 218: 209, 1991).
2) Rather than being formed from the termini of peptides, the telopeptides are generated by turns in the non-gly-X-Y regions (4, 6 in Fig. l) and stabilized by disulfide linkages; the location of cysteines is consistent with formation of disulfide bridges between the ends of 1 and 5, and start of 5 and the end of 7. Chou/Fasman algorithms predict alpha-helix at both turns. The distribution of lysine residues in these turns is characteristic of other telopeptides..
3) Sequences towards the amino terminus are not shown as being involved in folding as they presumably include a signal sequence and pro-collagen domain that are absent in the mature protein. It is not inconceivable that the putative pro-collagen peptidase target is the "Box A consensus" (Levy, et al., WBG 11(5): 36) in the center of the otherwise hypervariable region 2. "Box A" also contains a putative glycosylation site.
It has been suggested that the semi-dominant alleles of dpy-13 (e.g. e184 )might have products with aberrant collagen (triple helix?) formation but normal sites of interaction with other cuticle components, whereas the products of recessive alleles (e.g. e225 )might have normal collagen helix but an altered ability to interact with other components (von Mende and Riddle,WBG 11(4): 50). The molecular location of these defects is consistent with our folding model.
1. The size of collagen monomers calculated by these workers is not in accord with that predicted from C. elegans gene sequence data. We feel that the conditions used for their sedimentation studies (low concentrations of reducing agents) were conducive to cross-linking of monomers.
[See Figure 1]