Worm Breeder's Gazette 11(4): 90
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
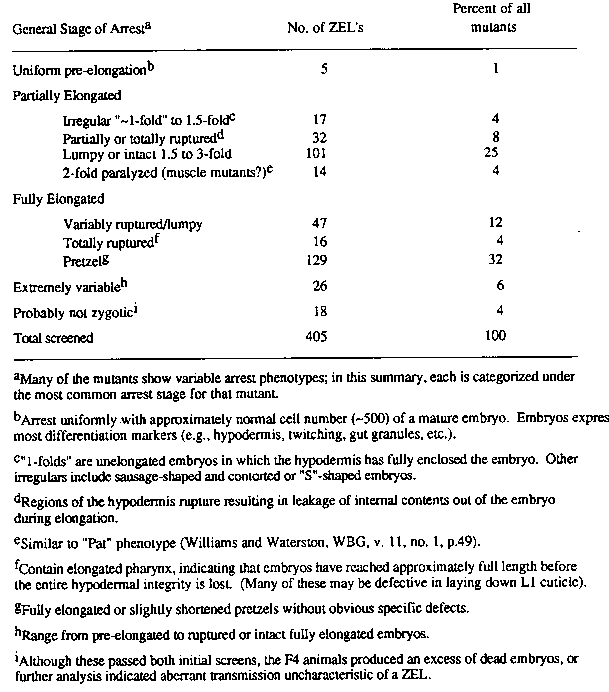
One way to identify essential genes likely to play controlling roles in embryonic development is to screen for zygotic embryonic lethal ( ZEL) mutations covered by a duplication or balancer. However, such screens limit one's search to a fraction of the genome, thereby preventing an assessment of a broad range of phenotypes. With the aim of analyzing a large proportion of all possible ZEL mutants in the worm, I have developed a procedure for screening the entire genome without the limitations imposed by balancers. In the screen, F1 self-progeny of mutagenized hermaphrodites are singled into microtiter wells and removed after 2 days of egg-laying. The F2 brood is then screened for ~25% arrested embryos 12 hours later. The screen is repeated on 6 F2 surviving siblings from each of the wells that contained 25% arrested embryos. Rescreened F2's that produce 25% dead eggs in the expected ratios are allowed to propagate to F4 broods of about 10,000 starved L1's, which are immediately frozen on the 35mm plates used in the follow-up screen. Up to 40% of the putative ZEL's identified in the primary screen behave aberrantly in the subsequent screen and are discarded, demonstrating the importance of the secondary screen. Initial attempts at this unbalanced screen were complicated by the finding that at standard levels of mutagenesis most wells contained some dead eggs (comprising from 1 to 100% of the F2's), and a high proportion of the 25% class failed to show transmission characteristic of a single locus ZEL. I found that by lowering the mutagenesis level the aberrant classes disappeared at a much greater rate than the ~25% class; at an optimized level, most of the mutants passing both screens are bona fide ZEL's. This screening procedure has allowed me to build up an extensive permanent collection of ZEL's. In each frozen plate there are enough worms to allow repeated sampling (~10 times) of many F4's without having to reisolate the lethal mutation. ~40% of the frozen worms are heterozygotes, providing sufficient fresh homozygous F5 embryos for screening by Nomarski, or with antibodies. To date, I've isolated 572 independent ZEL's from 15,800 F1's. I would like ultimately to approach saturation of the genome. Depending on the method of calculation, my current collection should have already identified on the average ~1-2 alleles per genomic ZEL locus. For the primary phenotypic screening, I've been examining the terminal phenotypes of all mutants by Nomarski. So far I've looked at 405 ZEL's and have carried out mapping, staining with antibodies, and lineage analysis on a selection of those with the most intriguing phenotypes. 11 ZEL's, selected on the basis of their interesting terminal phenotypes, mapped to 5 chromosomes as follows: LGI -- 2 mutants; LGII -- 4 mutants; LGIV --1 mutant; LGV -- 3 mutants; LGX --1 mutant. Therefore, as expected, the screening procedure works to identify ZEL genes broadly distributed over the entire genome. One mutation of interest mapped far out on the right arm of LGV -- a region that may never be covered by a balancer chromosome owing to its proximity to the presumptive pairing site; this illustrates one advantage of this screen over a balanced lethal screen. There are two cases in which pairs of mutants, selected on the basis of their similar unique phenotypes, mapped to similar regions (and are probably allelic), demonstrating that it is possible to identify multiple alleles of unique genes reproducibly based solely on their terminal phenotypes. The distribution of general arrest phenotypes for all 405 mutants is shown in the table. The most common arrest phenotype is a pretzel with no obvious defects; most such mutants are probably defective in 'housekeeping' genes. None of the mutants arrests uniformly with substantially less than the normal complement of cells (500) in mature wild-type embryos. The phenotypes of some of the mutants are particularly striking. One mutant (e2483 X) shows a (partial) block of gut differentiation, and arrests as an irregularly shaped, partially elongated embryo. Six mutants [e2503 II; ZE199 & ZE312, LGI (probably allelic); ZE200, LGIV; ZE372; ZE378] generally fail to undergo pharynx morphogenesis similarly to pha-1 mutants (Schnabel and Schnabel, Science, in press), but also show pleiotropic effects on elongation, arresting at some stage between the onset of elongation (i.e., the hypodermis encloses the embryo, but elongation hasn't begun) and the pretzel stage. These mutants generally lack a pharynx lumen, grinder, buccal cavity, and organized muscle fibers, but do have a basement membrane surrounding an under-developed pharynx. In several [ZE199 (and probably ZE312), ZE200, ZE372, and ZE378], expression of late pharynx antigens, such as myosin C and a gland-specific antigen are blocked; earlier antigens are generally unaffected. The defects in pharynx differentiation are not likely to be the result of a failure in elongation per se, since some mutants that don't initiate elongation nevertheless express late pharynx antigens (see accompanying article). Two allelic mutations ( e2501 II and ZE256, LGII) result in arrest as irregularly shaped partially elongated embryos with an abnormally large number (~15) of persistent cell deaths in the head, an incompletely formed pharynx and generally no discernible rectum. Finally, only five mutants arrested uniformly prior to the onset of elongation, resulting in embryos that lack hypodermis over most of their surfaces; this class of mutants is discussed more extensively in the accompanying article. Continued screening and in-depth analysis will allow me to assess more completely the range of developmental events during embryogenesis that are under control of the zygotic genome. A particularly interesting class of genes to me are those that control hypodermal fates and behavior, I believe many of these are likely to be identified among the classes of mutants that arrest a) prior to elongation (5 mutants), b) as partially elongated embryos with irregular shapes (17 mutants), and c) with loss of hypodermal integrity during elongation (32 mutants). Analysis of these mutants with antibodies to hypodermal antigens, and by lineaging, should allow me to identify genes required for specification and function of the hypodermis. [See Figure 1]