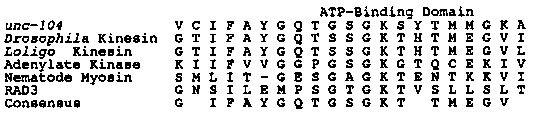Worm Breeder's Gazette 11(4): 67
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The unc-104 mutant defects include: 1) a reduced number of vesicles in the presynaptic region and their accumulation in the cell bodies, 2) a decrease in the volume of the synaptic region, 3) the reduction of presynaptic electron dense material, 4) the loss of neuromuscular junctions, and 5) improper growth of the muscle arms (Hall, D. H. et al., Soc. Neurosci. Abstr., 15, 1388 (1989) ) . In a search of Genbank (March 1990, v. 63) the unc-104 protein demonstrated homology to the ATPase and tubulin binding region of the kinesin heavy chain (46% identity in 240 amino acids), and a potential secondary structure similar to kinesin. The homology is strongest in the ATP-binding domain (see figure below). In other organisms, kinesin is believed to be involved in neuronal anterograde fast axonal transport and in microtubule-based movement in other cell types (for a review see McIntosh, J. R and Porter, M. E., J. Biol . Chem., 264 , 6001-6004 (1989)). Kinesin is a dimer of heavy chains (110-134 kDA), each associated with a light chain (60-80 kDa). The ATPase and microtubule-binding domains are located in the globular head near the heavy chain amino terminus, although in some kinesin- related proteins the 'motor' domain is at the carboxyl end. The unc- 104 protein (approximately 1600 amino acids) is larger than Drosophila kinesin (975 amino acids) and squid axon kinesin (967 amino acids). Further studies will be required to determine whether the unc-104 protein is functionally equivalent to kinesin. Because of the excellent genetics of the nematode, it should be a useful system for studying the role of kinesin-like proteins in axonal transport and synaptogenesis. [See Figure 1]