Worm Breeder's Gazette 11(4): 108
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
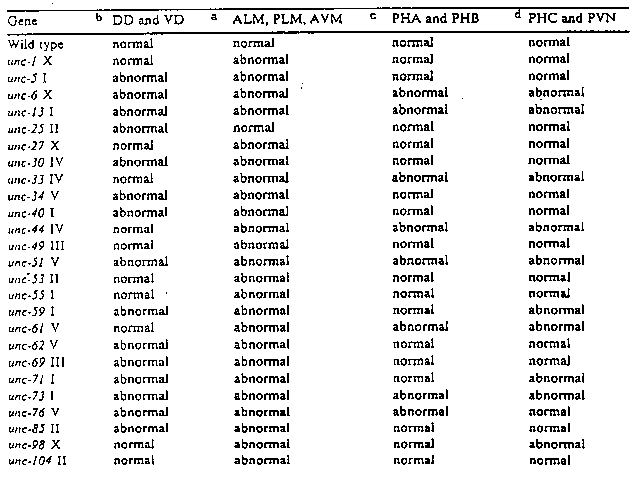
I have previously reported (e.g. CSH meetings 1985, 1987, 1989) several monoclonal and polyclonal antisera that can be used to specifically label different subsets of identified neurons in C. elegans. Here I wish to report a comparison of axonal guidance defects as observed in the set of unc mutants (Brenner, 1974) by immunocytochemical staining of whole animal squash preparations. For mutants in every gene several alleles (where available) were stained. I have included only those genes for which the penetrance of the axonal defect was at least 15%; e.g. unc-18 worms show abnormal touch cells, but it is not included here, since the defect is observed in only about 10% stained worms. [See Figure 1] Desai et al. (l988) have described mutants in eight of the above mentioned genes (unc-6, unc-33, unc-34, unc-40, unc-51, unc-71, unc-73, and unc-76) affected in the axonal guidance of HSN neurons. I have termed 'abnormal' to denote any deviation in neuronal cell body position, axonal outgrowth and or process placement, from the normal pattern observed in the wild type. Conclusions: Our results suggest that in C. elegans, sensory and motor neurons require the combined activity of a large number of genes: genes that affect the generation of migration of neurons and genes that axonal growth and process placement, i.e. the neural circuitry is a result of concerted activity of a group of genes that are required in more than one neuronal type. In addition, I find that these mutants show great variability in expressivity and penetrance of axonal defects, although the locomotory defects are very strong. It is likely, that some of the defects observed in axonal growth are caused by secondary defects in non-neural tissue, including hypodermis and muscle. Leakiness of a mutation may also cause variability of the axonal defect. Temperature sensitive alleles of these mutations may provide a correlation between behavioral alterations and changes in neural circuitry. Apparently there are very few genes that affect only one class of neurons selectively; even if such genes are discovered, the phenotype may simply reflect a differential requirement for the gene products. I would like to thank, J. Culotti, M. Chalfie, R. Durbin, E. Hedgecock, R. Horovitz, S. McIntire, J. Sulston, and J. White, for many useful discussions. I thank J. Culotti, R. Holmgren and J. Miwa for support and encouragement.