Worm Breeder's Gazette 11(1): 63
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
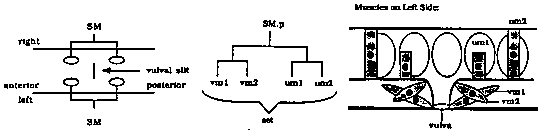
The hermaphrodite sex muscles, comprising two types of vulval (vm1 & vm2) and two types of uterine muscles (um1 & um2), are essential for egg laying. Each of the four SM.x myoblasts give rise to one set of muscle cells comprising one of each type of muscle cell. We have determined which of these cells are required for egg laying. [See Figure 1] Specific cells were killed by laser ablation in mid-to-late L3 hermaphrodite larvae and the kills were confirmed 1-4 hours afterwards by Nomarski microscopy. The vulval muscles were visualized in young adults by polarized light microscopy to determine if specific ablated cells were being replaced by others. The animals were then scored for their abilities to lay eggs. In general, ablation of specific cells resulted in the absence of the corresponding muscle(s), although occasionally ablated cells appeared to be replaced. However, animals in which replacement appeared to occur and those in which it did not behaved identically in their abilities to lay eggs. From this result we conclude that although the muscle attachments can sometimes rearrange, their functions are not fully interchangeable. To determine whether all four sets are required for egg laying, various combinations of the SM.x myoblasts were ablated. Animals missing one set are essentially wild-type, while those missing three or all four sets fail to lay any eggs and form bags of worms. Animals with two sets on the same lateral side, and those with an anterior set from one side and a posterior set from the other side lay eggs almost as well as unablated animals. In contrast, animals with only two anterior or only two posterior sets bloat with eggs. Thus, it appears that some cells from at least one anterior and one posterior set are required for effective egg laying. We also ablated individual types of muscles in all of the sets, demonstrating that the vulval muscles are necessary and sufficient for normal egg laying and that the vm2's play a more important role than the vm1's. Animals missing all eight uterine muscles lay eggs apparently normally, while those missing all eight vulval muscles fail to lay any eggs. Ablation of the four vm2's results in egg-laying defective animals that form bags of worms, while ablation of the four vm1's results in animals that lay eggs almost as well as unablated animals. The vm2's are the only egg laying-muscles that receive direct innervation from the HSN neurons that drive egg laying. While it is possible that the defect caused by ablation of the vm2's is due to the inability for the remaining muscles to be stimulated to contract, the majority of animals missing the HSN's eventually lay their eggs and do not form bags of worms. Thus, the role of the vm2's in opening the uterine/vulval connection is probably also important in egg laying even in the absence of innervation by the HSN's.