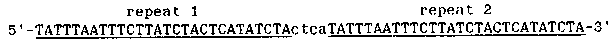Worm Breeder's Gazette 10(3): 16
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The tra-2 gene product is required to direct female development in C. elegans. In the hermaphrodite germline, female development must be transiently repressed to allow spermatogenesis. The product of the fog-2 gene is required for spermatogenesis only in hermaphrodites and is postulated to be involved in this transient inhibition of female development (1). Gain-of-function mutations have been isolated in tra- 2 which produce XX females rather than self-fertile hermaphrodites(1,2) . The existence of these tra-2(gf) alleles suggests that transient male development in the hermaphrodite germline may be the result of modulation of tra-2 activity. We have begun a molecular characterization of these tra-2(gf) alleles to understand the mechanism of this regulation. tra-2 spans a region of about 10kb as defined by Tc1 insertions and small deletions in mutant alleles. Major transcripts of approximately 5kb, 2.8kb, and 1.4kb are detected on Northern blots of adult hermaphrodite polyA+ RNA using probes spanning this 10kb region. These three RNAs are transcribed from the same DNA strand and contain common sequences. Only the 5kb band is altered in mobility in any of the tra-2 gain-of-function or loss-of-function alleles tested. The 5kb hand is composed of at least two co-migrating species. We have not yet determined the differences between the 5kb transcripts. Alterations in the DNA near the 3' end of the tra-2 transcription unit have been identified by Southern blot in 6 tra-2(gf) alleles. Small deletions are present in 4 alleles (q122gf, q244gf, e2046gf, and e2020gf). Tc1 insertions are present in 2 alleles (q101gf and q103gf ). The remaining 6 tra-2(gf) alleles (q179gf, e1939gf, e1940gf, e1941gf, e2019gf, and e2021gf ) do not contain lesions which are detectable on Southern blots . The restriction fragment to which the gain-of-function mutations map contains two identical copies of a 28bp sequence which form a direct repeat separated by 4bp (see Fig. 1). This direct repeat lies 52bp upstream from an AATAAA putative polyadenylation signal. Partial sequence data from a 1.3kb tra-2 cDNA clone indicates the direct repeat lies in the 3' untranslated region of an RNA which terminates at this poly-adenylation site. The restriction fragment containing the deletions was cloned from tra-2 q122gf, q1244gf, e2046gf, and e2020gf using Polymerase Chain Reaction (PCR) to amplify this fragment from genomic DNA. Sequence analysis of these clones indicates that q122gf, q214gf, and e2046gf contain identical 32bp deletions that remove one copy of the 28bp repeat and the 4bp spacer between the repeats. e2020gf contains a 108bp deletion that removes both copies of the direct repeat. Six cIones isolated from e2020gr also contain a point mutation 100bp upstream from the 108bp deletion. (This point mutation creates an MboI restriction site; preliminary evidence indicates that this novel MboI site is present in e2020gf genomic DNA.) It is not yet known whether this point mutation is present in the 3'-UTR or protein coding sequence. q122gf, q244gf, and e2046gf are phenotypically identical, consistent with the identical DNA sequence of the mutant alleles. e2020gf is phenotypically different from q122gf etc. As described by Doniach(2), e2020gf is partially defective in tra-2 regulation in XOs as well as being effective in the transient modulation of feminizing activity in hermaphrodites. This suggests that her-1 regulation of tra-2 activity in XOs may also be affected by sequences at the 3' end of the tra-2 gene. The 6 tra-2(gf) alleles that appear to be point mutations are weak relative to e2020gf or q122gf etc. as well as exhibiting hypomorphic character when placed in trans to a tra-2(lf) allele (2). One model for the mechanism of transient inhibition of female development in the hermaphrodite germline might have been that the fog- 2 gene product binds to the direct repeat at the 3' end of tra-2 ( either to DNA or RNA) and transiently inhibits tra-2 activity. Schedl and Kimble (1) showed that as homozygous for tra-2q122gf, q244gf, and e2046gf can still respond to regulation by the fog-2 gene product. However, the fog-2 regulation of these tra-2(gf) alleles might have occured through the remaining copy of the 28bp repeat. We tested whether animals homozygous for e2020gf, which deletes both copies of the 28bp repeat, can respond to the fog-2 gene product. fem-3(q95gf) partially suppresses tra-2(e2020gf) leading to hermaphrodite rather than female development. Our results indicate that the fog-2 gene product is required for spermatogenesis in tra-2(e2020gf);fem-3(q95gf) hermaphrodites. This result shows that the fog-2 product does not require any part of the direct repeat to promote spermatogenesis in the hermaphrodite germline. The molecular defect in these tra-2 gain-of-function alleles indicates some regulatory role for the direct repeat. The sequence of these repeats is AT-rich (80%) and shares limited similarity with AU- rich sequences responsible for rapid mRNA turnover found in the 3'-UTR of some mammalian genes (3). However, the level at which tra-2 activity is regulated in C. elegans is unknown. Interestingly, the mutations found in fem-3(gf) alleles also map to the 3'-UTR of the gene (see the abstract by Ahringer et al., this issue). This may suggest some similarity between the mechanism of regulation of tra-2 and fem-3.