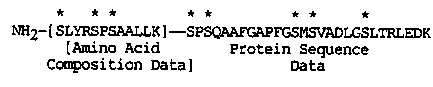Worm Breeder's Gazette 10(3): 108
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
In the vol.10, number 2 edition of The Wormbreeder's Gazette, we reported that paramyosin is phosphorylated in vitro under low salt conditions by an endogenous kinase in a reaction that is Ca++ independent. Thin layer chromatography indicated that paramyosin contained phosphoserine. Phosphate assays suggested that in vivo paramyosin had 1.8 moles of phosphate per mole of paramyosin. To determine the sites of phosphorylation, in vitro phosphorylated paramyosin was digested by NTCB. A 15,000 Da fragment was phosphorylated which corresponded to the N-terminal fragment according to Hiro Kagawa's sequence data. To obtain a more precise localization, in vitro labelled paramyosin was digested by endoproteinase-Lys-C. 2 labelled HPLC fractions were obtained and subjected to protein sequencing. One however yielded no sequence, and since the N-terminus of paramyosin is known to be blocked, probably by acetylation, we guessed that this peptide included the blocked N-terminus. Amino acid composition of the peptide was determined. The second peptide was sequenced with no difficulty. This sequence and the amino acid composition data of the blocked peptide were compared with H. Kagawa's DNA sequence of the paramyosin gene, unc-15. They appear to arise from, and together define the N-terminus of paramyosin. [See Figure 1] With three prolines, two glycines, and eight serines(*), the N- terminal region that we have determined is decidedly not alpha-helical coiled-coil in structure and is substantially different then the remainder of C. elegans paramyosin (Kagawa's sequence) and myosin rod sequences. It would be interesting to determine what role the phosphorylation of the non-helical N-terminus of paramyosin plays in the interaction between paramyosin and other proteins in C. elegans' thick filament formation and function.