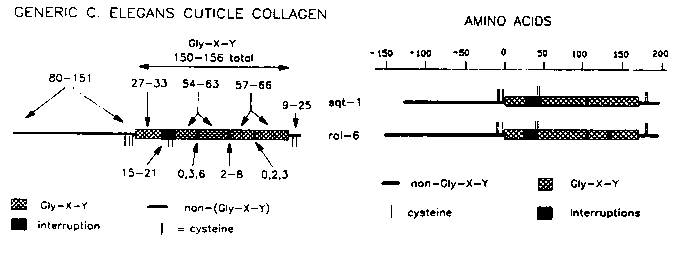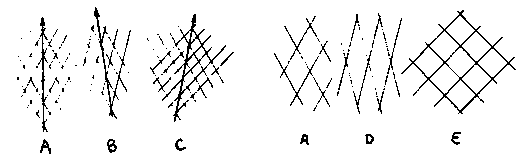Worm Breeder's Gazette 10(2): 75
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
rol-6 and sqt-1 encode collagens that are very similar in structure to the other sequenced collagens. The sequences of the wild-type alleles of rol-6 and sqt-1 have been determined, and a diagram of the predicted protein products is shown below. Also, a diagram of the 'generic' cuticle collagen structure, derived from the sequences of nine other collagens (col's 1,2,6,7,8,12,13,14 and 19), is shown. The overall structures of rol-6 and sqt-1 fit the 'generic' structure. It is apparent that the overall structures of collagens that can cause a Roller phenotype are not obviously unique. rol-6 has four short (6-9 amino acid) Gly-X-Y repeats, three of which are found in front of the first major Gly-X-Y block, and the other in the carboxy-tail. These Gly-X-Y repeats are not found in the other collagen genes, but are probably too short to produce stable triple-helical structures. rol-6 and sqt-1 are more similar to each other than to the other sequenced collagens. Comparisons of these eleven collagens in the regions around the conserved cysteine residues, in the first Gly-X-Y interruption, and in the carboxy-tail, indicate that in these regions rol-6 and sqt-1 have more amino acid sequence similarity to each other than to the other nine collagens. The overall level of amino acid similarity between rol-6 and sqt-1, however, is not greater than between some other pairs of genes (e.g., 1 and 2, 8 and 19, 12 and 13). These regions of similarity between rol-6 and sqt-1 could be functionally significant and define critical amino acids in collagens that are capable of producing Roller phenotypes. However, it is also possible that the similarity is the result of rol-6 and sqt-1 having arisen from a relatively recent gene duplication event, since the two genes are just 1.4 mu apart on LG II. In this case, the similarity may reflect ancestry rather than function. col-6 is also located on LG II (less than 1 mu to the left of rol-6) but does not have a high degree of amino acid similarity to rol-6 or sqt-1 in the regions analyzed. [See Figure 1] The Tc1 insertion in the CH1 strain is within the coding region of sqt-1. We have mapped the location of the Tc1 insertion in CH1 sqt-1( sc143 cg1) to be 16 to 17 amino acids in from the putative translation start site. This region is hydrophobic and likely to be part of the signal sequence. CH1 has the same genetic properties as the putative sqt-1 nulls that were described by M. Kusch and B. Edgar, supporting the view that the null phenotype for sqt-1 appears wild-type. A sqt-1 left-roller strain and the CH1 putative null strain have disrupted cuticle ultrastructure. We have some preliminary results of SEM analysis of the left roller allele sc143 and its Tc1 insertion derivative CH1 (sc143 cg1). SEM analysis of ripped cuticles reveal that the struts and fiber layer(s?) are disorganized in sc143 cuticles. In N2 the upper fiber layer is oriented at approx. 60 degrees from the long axis of the animal, while in sc143 the fiber layer appears to be oriented almost perpendicular to the long axis. These results are consistent with TEM pictures of sqt-1(sc13) left-roller cuticles produced by Bob Edgar's group. Surprisingly, the struts and fiber layer(s) of CH1 cuticles are more severely disrupted than those of sc143, even though the dissecting scope phenotype of CH1 is essentially wild-type. The fiber layer in CH1 doesn't even appear fibrous. This result indicates that the sqt-1 gene product is required for normal cuticular ultrastructure and its absence is not compensated for by another member of the collagen gene family. A simple model for cuticle morphology. The basal layer of the cuticle contains two apposed fiber layers that are oriented at equivalent but opposite angles to the long axis of the animal. Viewed from above the fibers in these layers produce a set of parallelograms between them. If one draws a line through the vertices of these parallelograms it will run parallel to the long axis of the animal ( see A below). If the angle of one of the layers is increased or decreased, a line through the vertices will run at an angle to the long axis producing either a right or left-handed helix - the Roller phenotype (B and C). If both layers are turned closer to the long axis a longer, narrower parallelogram results - the Long phenotype (D). If both layers are turned further from the long axis a shorter, wider parallelogram is produced - the Dumpy phenotype (E). Thus, changes in orientation of the fiber layers could be the basis for some mutants that affect morphology. Since sqt-1 mutations can affect the fiber layers, their orientation may at least partially be controlled by their structural components. The possible importance of the fiber layers in cuticle morphology was also proposed by Cox et al.(Genetics 95:317, 1980). As with most simple models this one is probably incorrect, but it may be useful for thinking about the complex genetics of some morphological mutants. [See Figure 2]