Worm Breeder's Gazette 10(2): 67
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
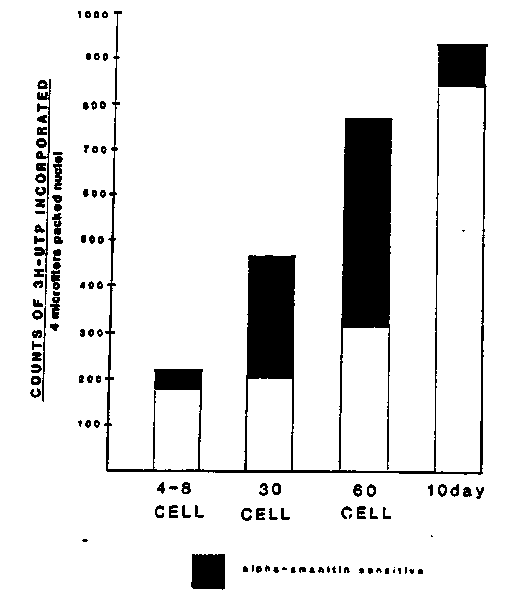
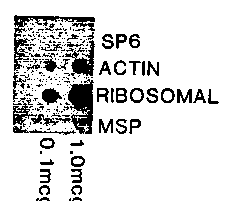
We have exploited some of the unique features of embryos of the parasitic roundworm, Ascaris des to observe transcriptional patterns in nematode early embryogenesis. In contrast to the free-living nematode Caenorhabditis ble to obtain large synchronous populations of Ascaris embryos at various stages of development. It has been reported in C. elegans that transcription is detectable at approximately the 100-cell stage. [Hecht, et al., (1981). Dev. Biol. 83:374-379]. More recent work in C. elegans suggests that transcription is occurring at or before the 30-cell stage. [Schauer, et al., WBG 10:1, 72-73.]. Our laboratory has recently reported that Ascaris embryos in the 5-6th cleavage stage are actively transcribing. [Dalley, et al., WBG 10:1, 76.]. From staged embryos we have isolated nuclei to determine run-on transcription characteristics. Initial studies performed on nuclei isolated by the method of Dixon et al. [WBG 9:3, 73-74.] resulted in contamination of the embryonic preparations with mitochondria, as seen by microscopic examination after diamidinophenylindole (DAPI) staining, as well as a strong hybridization signal using a mitochondrial probe. Subsequent purification of nuclei on 50% Percoll gradients has yielded about a 100-fold visual reduction in mitochondria, although a hybridization signal can still be detected. Run-on transcription assays were performed on nuclei isolated from staged embryos of 4-8 cell, 24-30 cell, ~60 cell, and 10 day embryos (about 600 cells). Parallel assays were carried out with and without alpha-amanitin present at 1 g/ml a level which inhibits RNA polymerase II activity. The results are shown on Fig. 1 as counts of [3H]-UTP incorporated per 10 l reaction. Total levels of incorporation were typically 10-35 fold over background, which has been subtracted from each of the values shown. The darkened portion of each bar represents the alpha-amanitin sensitive (RNA polymerase II) fraction of total transcription. We consistently detect RNA polymerase II activity as early as the 4-8 cell stage and by the 30 cell stage active mRNA transcription is occurring. When DNA content of preparations was measured by fluorimetric assay the highest level of incorporation per pg DNA was at the 30 cell stage. Similar transcription reactions were carried out with [32P]-UTP, with the labelled RNAs used to probe duplicate dot blots containing cold DNA probes. Results from the 60-cell stage showed no hybridization to the SP6 vector or the Ascaris sperm- specific MSP cDNA. Ascaris ribosomal and actin probes showed significant hybridization (Fig. 2). When the labelling was carried out in the presence of 1 I/ml alpha-amanitin, the actin signal was eliminated, while the ribosomal signal was retained as expected. This shows that we are able to detect specific mRNA transcription occurring in staged embryos. Similar studies are being carried out with each of the mentioned cell stages to establish if specific messages, i.e., actin for now, can be detected at the 4-8 cell stage. We propose that differential library screening using staged early vs. Iate RNA populations will yield transcripts unique to the early embryo. The following two references have been valuable for run-on transcriptions: J. R. Nevins, Methods in Enzymology, (1987), 172:235-241. M. E. Greenberg, Current Protocols in Molecular Biology, (1987), 4. 10.1-4.10. 7