Worm Breeder's Gazette 10(2): 53
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
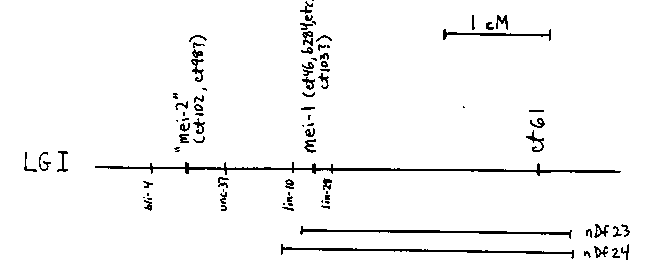
We have previously described the interactions between alleles of mei- 1(I) and the mutations ct46(I) and ct61(I) [Mains et al., WBG 10(1) :102]. The latter two mutations produce gain-of-function 'poison' gene products that result in dominant, temperature-sensitive maternal- effect lethality. The mutations are linked, but represent different loci separated by 2 cM. Mutations of mei-1 show nonconditional recessive maternal-effect lethality resulting from a failure of meiosis; the canonical allele (b284) was isolated in a screen for maternal-effect lethals [Sprunger and Kemphues, WBG 10(1):100]. This mutation is an efficient trans-dominant suppressor of both ct46 and ct61. Previously, we had isolated twelve additional alleles of mei-1 as suppressors of ct46. Here we show that ct46 is an allele of mei-1. One of these suppressor mutations, ct99, was isolated in cis to ct46. The eggs from ct46ct99/+ + mothers show wild-type levels of hatching compared to l% for the eggs of ct46/+ mothers. ct46ct99 shows recessive nonconditional maternal effect lethality and fails to complement mei-1(b284). ct46ct99/ct46+ shows no trans-suppression of ct46. An additional suppressor mutation, isolated as ct46ct101, shows identical behavior. Therefore, ct101 and ct99 are cis-dominant suppressors of ct46 apparently resulting from intragenic loss-of- function reversion of the ct46 gain-of-function poison product. Thus, ct46 is an allele of mei-1. Consistent with this interpretation, mutations in mei-1 map <0.01 cM from ct46. These revertants occurred at a high frequency: 2/5500 after EMS mutagenesis, again implying that they are loss-of-function mutations. While the above interpretation of the cis-dominant suppression of ct46 by ct99 and ct101 is straightforward, the majority of mei-1 alleles, for example ct82, shows the surprising property of also being trans-dominant. As reported previously [Mains et al., op. cit.], 92% of the eggs from ct46ct82/ct46+ mothers hatch. Twelve alleles of mei- 1 (defined by the failure to complement for the recessive maternal effect lethality) show this property, and the trans-suppression ranges from 28-92%. Like ct99 and ct101, these alleles appear to represent general loss-of-function rather than specific gain-of-function mutations that compensate for the ct46 lesion. They were isolated at a very high frequency after EMS mutagenesis (11/5500); moreover, the canonical allele b284, which was isolated in the absence of ct46 (see above), is also an efficient trans-suppressor (77% of the eggs from ct46+/+b284 hatch). Unlike ct99 and ct101, these mutations are also trans-dominant suppressors of ct61. How can we reconcile these two classes of apparent loss-of-function alleles of mei-1, one which suppresses ct46 only in cis and the other which can suppress ct46 (and ct61) in cis or trans? It seems likely that the cis-only suppressors (ct99 and ct101) are true null mutations (e.g., causing absence of all gene-product functions) while the trans suppression (ct82, b284, etc.) is mediated by the presence of a partially defective gene product. Deficiencies for the region (nDf23 and nDf24) show no trans-suppression, consistent with the interpretation that the cis only suppressors represent the null phenotype (although this interpretation is complicated by the fact that the deficiencies tested remove the ct61 locus in addition to mei- 1). One possibility consistent with the evidence would be that a partially defective mei-1 product can form a complex with the poison ct46 (or ct61) gene product and thereby inactivate it, whereas a completely defective (or missing) mei-1 product resulting from a null mutation cannot. It should be noted that Kusch and Edgar [Genetics 113: 621 (1986)] saw a similar pattern of interactions for several genes affecting the cuticle (i.e. loss-of-function mutations, but not true nulls, could suppress dominant gain-of-function mutations). We may have also identified yet another class of mei-1 alleles. The mutation ct103(I) is a trans-dominant suppressor of both ct46 and ct61, but unlike the previously described mei-1 alleles shows no recessive maternal-effect lethality. It complements other mei-1 alleles. However, it maps between lin-10 and lin-28(I), the closest markers that flank mei-1.Therefore, we have an allelic series of mei-1: (1) ct46, a dominant gain-of-function poison, (2) ct99 and ct101, putative null alleles that are cis-dominant suppressors of ct46 and recessive maternal-effect lethals, (3) ct82, b284 (and 9 others), trans-dominant suppressors of ct46 and ct61 and recessive maternal effect lethals, and (4) possibly ct103, a trans-dominant suppressor of ct46 and ct61 with no recessive phenotype. We have identified a mutation at another locus, ct102(I), which is also a trans-dominant suppressor of both ct46 and ct61. It shows recessive maternal-effect lethality with defects that closely resemble those of mei-1 mutants (Sprunger and Kemphues, op. cit.). However, ct102 complements recessive alleles of mei-1 and maps to a different interval, between bli-4 and unc-37. This locus will likely be designated 'mei-2'. Finally, a second mutation that maps to this same interval, ct98, is also a trans-dominant suppressor of ct46 and ct61, but complements ct102 and shows no recessive maternal-effect lethality. Therefore, this could be a viable allele of 'mei-2'.[See Figure 1]