Worm Breeder's Gazette 10(2): 50
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
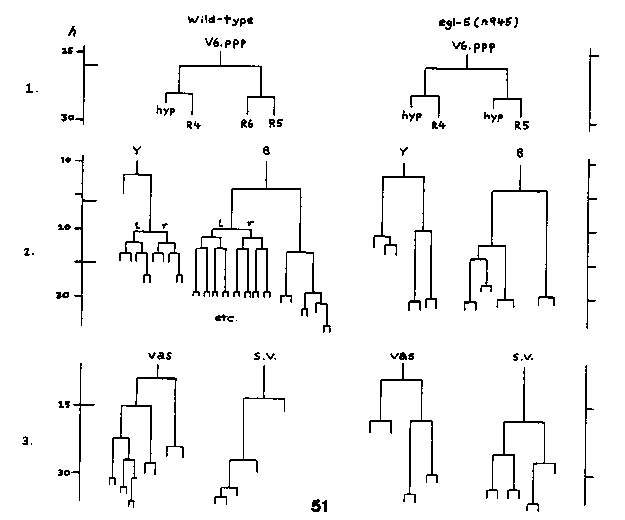
Mutations in the gene egl-5 III have highly pleiotropic effects. Hermaphrodites are Egl as a result of defective HSN migration. A number of observations suggest that this is due to incorrectly specified HSN identity (Chand Desai, pc). Hermaphrodites mutant for the candidate null allele n945am and 5 others (including the reference allele n486) are also Unc (spontaneous backward coiler, ventrally or dorsally) and are tail Mec, both from L1 onwards. Males mutant for any of these 6 alleles are severely abnormal (Mab). An exceptional allele, n1439, which has the lowest Egl penetrance (92%), and which is not itself Unc, Mec or Mab, fails to complement the others for Egl but complements them for Unc, Mec and Mab (Desai, pc). To discover the basis for the Mab phenotype, I have examined the postembryonic lineages of egl-5;him-5(e1490) males. The account below is based on 4 n486 males and 3 n945 males lineaged from early L1 to the L3 moult or later, as well as several partial lineages for both genotypes. Ventral hypodermis: In wild-type males, P(9-11).p constitute the preanal equivalence group, which generates the hook. The three cells in the group undergo lineages designated 3 , 2 , or 1 respectively, based on the replacement hierarchy seen in ablation experiments; P12.p divides in L1 and does not participate in the equivalence group. In n486 and n945 males, the whole preanal equivalence group is effectively shifted posteriorly: P12.p fails to divide in L1, but instead survives until L3 and undergoes a 1 lineage; P11.p has a 2 fate and P.10.p is 3 . This is seen in all animals, although there is some slippage: P9.p may still participate as a 3 lineage, and P10.p may execute a lineage intermediate between 3 and 2 . P12.aap divides in late L3, implying that the transformation is at the level of the P cells themselves rather than their posterior daughters. Preanal cytoplasmic retraction begins in L4, but no recognizable hook is made. A similar transformation is seen in let-23 males (Paul Sternberg, pc). P12.p fails to divide in egl-5 hermaphrodites as well. Lateral hypodermis: Lateral hypodermal lineages are wildtype except for one intriguing blip: the division of V6.pppp is unequal ( resembling the other V ray precursor parents), instead of being equal ( see Fig. 1). V6.ppppa, instead of giving rise to R6, is hypodermal. A plausible tail seam is made but its 5 nuclei fail to migrate anteriorly in L4. Tail cytoplasmic retraction begins normally, but there is no ventral retraction: a vestigial fan with 8 ray papillae on each side is made. Phasmid traces and T rays are always present. B, Y, U, F & K: The lineages of all the four male-specific blast cells (B, Y, U and F) are abnormal. None of the structures formed by the descendants of these cells (spicules, proctodeum, etc.) is made. All 5 cells are distinctly smaller than normal in L1. The first divisions of B and Y are on time but later divisions have abnormal axes and timing. Typical B and Y lineages in n945 are shown (Fig. 2) : the patterns are quite reproducible. The normal symmetrical (left- right) divisions are not observed in B, Y, U or F, although the latter two may divide in L3. K never divides in either sex in n945, although in n486 it can execute a proliferative lineage like that of Y above, i. e.,with a round of divisions at the end of L1, L2 and L3. Somatic gonad: The male somatic gonad is grossly abnormal, as a result of delays and defects in the late somatic gonad lineages. These cause gonad growth to grind to a halt during L3, when the reflexed arm is about half as long as the distal arm. The linker cell (which appears normal) subsequently breaks free of the rest of the gonad (it sometimes takes a couple of cells with it) and ploughs onwards, ending up somewhere between the distal tip cell and the cloaca. The abortive somatic gonad still manages to form a sac-like structure into which a seemingly normal germline proliferates. Pockets of spermatogenesis break out at the correct time; the gonad may burst, leaking sperm into the body cavity. The lineage defects are variable: representative ones are shown (Fig. 3). Both vas deferens and seminal vesicle lineages (of both Z1 and Z4) are abnormal. Usually the first round of vas divisions is OK, but thereafter chaos ensues. Hermaphrodite somatic gonads look completely wild- type. Occasionally (<5%?) QL migrates anteriorly (like QR). Other embryonic and postembryonic migrations (excepting the HSN) appear normal. M is wildtype at least until the divisions of the SM grand- daughters; 10/10 n945 males had dorsal coelomocytes (SM2L.pp). n945am is believed to be a null allele of egl-5 because the Egl does not get worse in n945/nDf16 (Desai, pc). To check this conclusion with respect to the Mab I have lineaged 3 n945/nDf16 males fully (and several partially). The ventral hypodermal defects do not get worse in the deficiency het. The lateral hypodermis is sometimes more disorganised. The somatic gonad defects may be slightly worse (in 2 animals the somatic primordium was disorganised from L1). The ectoblast lineages are difficult to interpret, but don't seem much worse. Therefore n945 may be null. However, none of the extant egl-5 alleles derives from a complementation screen in which lethal or sterile null alleles could have been picked up. The pleiotropic effects of egl-5 mutations are a puzzle. Some of the defects may reflect a local role for egl-5 product in the preanal or rectal region (HSNs are born here, near the PLMs). Several DA,DB and DD motoneurons and numerous interneurons also arise in the vicinity during embryogenesis, and defects in these might give rise to a backward coiler phenotype. It is difficult to fit the male somatic gonad defects into such a picture. Thanks to Chand Desai for sending egl-5 strains and information.