Worm Breeder's Gazette 10(2): 42
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
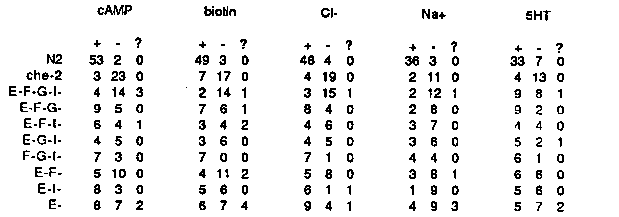
C. elegans chemotaxes to Na+ and Cl- ions, and several amino acids ( Ward, PNAS 70:817 (1973)). We are interested in studying this behavior at the cellular and genetic levels. Single animals are tested in a chemotaxis assay similar to one described by Ward. A gradient of an attractant is established by cutting a ~50 I (6 mM) plug out of a 2% agar plate, soaking the plug in attractant, replacing it, and allowing the attractant to diffuse for 12-24 hours. A single animal is placed on the plate, removed an hour later, and its tracks observed. An animal that arrives at the peak of the gradient more times than it arrives at a control agar plug is scored as positive (see table below for the behavior of control animals). Usually a single animal will be tested for its response to six attractants, though chemotaxis-defective animals are lost sometimes. Anyone interested in this assay or similar population assays should call to get more details about things that seem to make it work well. To define the cells involved in chemotaxis, we are killing neurons using a laser microbeam. The cells are killed at the L1 stage, and the animals tested for their behavior as young adults (0-36 hours after the last molt). The first cells we examined were the putative chemosensory neurons, i.e. those with endings exposed to the environment. There are nine classes of putative chemosensory cells in the head (22 cells total) in two types of sensillum, the amphid and the inner labial sensillum. The phasmids, in the tail, were shown by Ward to be unnecessary for chemotaxis; we have confirmed his result by finding that lin-17 mutants, which have defective phasmids, are wild- type for chemotaxis. Preliminary experiments suggested that the amphid contained cells necessary for chemotaxis. Exploratory kills within the amphid indicated that the only cell whose death reproducibly leads to ( partially) defective chemotaxis was ASE. It appears that several other chemosensory cells (some or all of ADF, ASG, and ASI) are also involved in chemotaxis. The data are summarized in the table below. [See Figure 1] Each entry in this table indicates the number of independent animals that gave a positive response (+), the number of animals that gave a negative response (-), and the number of animals that gave contradictory results in two or more assays with the same attractant (? ). Thus for N2, 55 animals were tested for their response to cAMP in single animal assays, and 53 gave a positive response. che-2(e1033) was used as a negative control. All chemosensory neurons are grossly defective in this strain, so the fraction of positive responses should give a good estimate of the fraction of fake positive results that will come out of the assay as scored. Biotin and serotonin (SHT) are 'new' attractants that we identified in a screen of chemicals. Given that the range of the assay is 12-29% positive for che-2 and 83-96% positive for N2, the numbers for the three-cell-kills are too small to be conclusive in some cases. However, the quadruple ASE ADF ASG ASI kill is very defective in its response to cAMP, biotin, Cl-, and Na+. The data suggest that these responses are mediated at least in part by the same group of redundant cells (we are still testing alternative explanations of the data). The apparent residual 5HT response of ASE- ADF- ASG- ASI- animals is being examined. Ever mindful of the possible problems with laser experiments, we have confirmed many kills of subsets of the chemosensory neurons by an independent assay. As described by Hedgecock et al., Dev. Biol. 111:158 (1985), six of the cells in each amphid take up FITC efficiently through their exposed endings. This observation allows us to confirm that cells whose nuclei have been killed are functionally defective, i.e. fail to take up FITC. Killing the amphid sheath cell may be a means of reducing the function of most amphidial cells (see Thomas and Horvitz, WBG 10:1 p. 89 (1987)). Amphid sheath-killed animals are partially defective in chemotaxis, but not so defective as animals in which ASE, ADF, ASG, and ASI are killed; for example, 7/12 amphid sheath-killed animals respond to Cl-, as compared with 4/19 E-F-G-I- animals. FITC fills of animals in which the amphid sheath cells have been killed showed that about a third of those animals still contain 1-3 (out of 12) dye- filling cells. If only one or two of the eight cells identified above need be functional for chemotaxis, this observation probably explains the residual chemotactic responses present in sheath-killed animals. In the course of these experiments, we noticed that when most of the amphid sensory cells are killed in a single animal (the best guess so far is the combination of ADF, ASG, ASI, ASJ, and ADL), the individual laser-ablated animals become dauer larvae constitutively on bacterial lawns at 20 C. A few of these dauers have been maintained for a week or more; they never recover to form adults. It may be that some or all of these five cells receive the food and pheromone signals that regulate dauer formation. When ADF, ASG, ASI, ASJ, and ADL were killed in daf-10(e1387) animals, 2/4 animals developed into dauers on bacterial lawns. This suggests that the dauer-defective lesion in daf- 10 is bypassed by these neuronal kills. N2 animals in which the combination of ADF, ASG, and ASI were killed took about a day longer to grow to adulthood than other animals. These animals appear to go through an L2-dauer stage (Golden and Riddle, Dev. Biol. 102:368 (1984)) instead of the normal L2 stage, suggesting a partial loss of food-sensing ability in these animals.