Worm Breeder's Gazette 10(2): 32
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
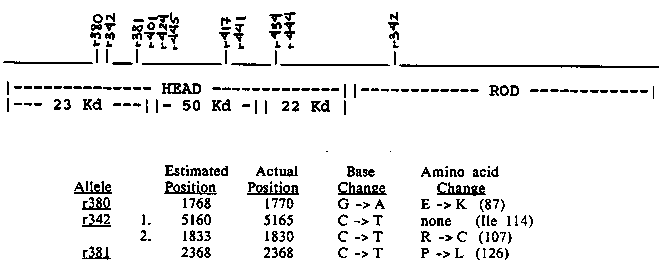
We are investigating the molecular basis of dominant antimorphic mutations at the unc-54 locus. These dominant-uncoordinated, recessive lethal mutations disrupt the assembly of thick filaments in the body wall muscle. By genetic and ultrastructural criteria, the mutant gene product interacts with both wild-type myosin heavy chain B, the unc-54 gene product, and myosin heavy chain A, the minor heavy chain class found in body wall muscle thick filaments. The strong disruptive effect is mediated by very low levels of full length protein - depending on the allele the amount ranges from less than 2% to approximately 20% of wild-type levels. Following EMS mutagenesis, these antimorphic missense alleles arise at a frequency equal to that of recessive null alleles at the unc-54 locus. To determine what portions of the myosin heavy chain are affected by these mutations, we have employed a technique developed by Myers et al. (Science 230: 1242, 1985) for detecting single base changes in genomic DNA. Base changes have been localized for 10 independently isolated EMS-induced alleles; all 10 show changes in the region of the gene encoding the globular head. [See Figure 1] One allele, unc-54(r342), carries 2 separate point mutations - one in the head and one in the rod portion of the gene. Both mutations have been sequenced and only the one located in the head creates an amino acid substitution. We have used the polymerase chain reaction ( PCR) technique (Saiki et al., Science 239: 487, 1988) to amplify and then sequence without cloning the affected region from mutant genomic DNA. Mutant sequences confirm the accuracy of mismatch positions estimated by the Myers technique. The head mutations sequenced so far create non-conservative substitutions for amino acids that are highly conserved among many myosin heavy chain sequences (including the yeast heavy chain). Thus we have mutationally identified parts of the globular head that are important in thick filament assembly. [See Figure 1]