Worm Breeder's Gazette 10(2): 25
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
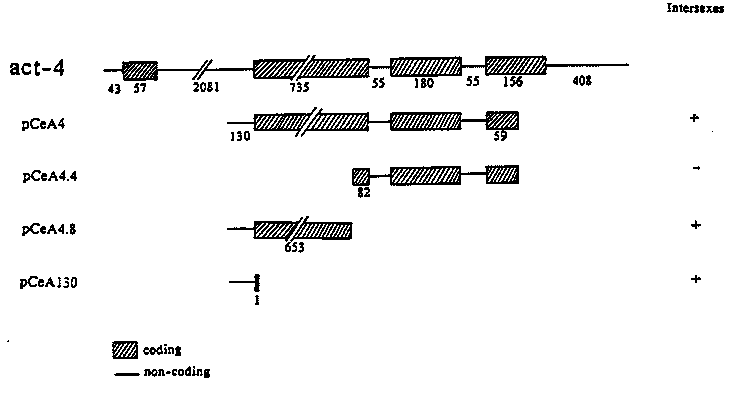
In the last WBG (10(1): 62-63), we described our initial experiments to define the molecular basis of the X-linked signal for sex determination in C. elegans. To summarize, cloned X-linked sequences were injected into the distal arm of the gonad of tetraploid hermaphrodites. The injected animals were mated to diploid males heterozygous for the X-chromosome duplication mnDp8 to produce 3A;2X cross progeny. In the absence of injected DNA, the 3A;2X + mnDp8 progeny are males (Madl and Herman, Genetics 93: 393-402, 1979). Injection of the act-4 plasmid pCeA4 (provided by Mike Krause) led to intersexual development of 3A;2X + Dp progeny. The intersexes are of a mosaic type in which nearly normal structures of both sexes are observed in the same animal. Injection of vector sequences, lambda DNA, or the autosomal gene act-3 had no effect on sex determination. We concluded that pCeA4 contained a sequence or sequences that are involved in assessment of the X/A ratio. By testing subclones of pCeA4 we find that the transforming activity of pCeA4 resides in the 5' 783 bp of the plasmid (plasmid pCeA4.8, see figure); no detectable feminizing activity could be found in the remainder (plasmid pCeA4.4) by this assay. Injection of a second subclone, pCeA130, which contains only 130 bp of the large intron and 1 bp from the coding region, also led to production of intersexual progeny. The transformation frequencies observed for all plasmids which give positive results are quite high: approximately one of every four fertile injected hermaphrodites produces intersexual offspring and about one-third of the 3A;2X + Dp offspring of such animals are intersexes. We are currently using exonuclease III/S1 deletions of pCeA130 to determine the minimal sequence required to obtain transformation. Initial results indicate that the insert retains feminizing activity after deletion of as much as 70 base pairs from the 5' end. Thus we have identified a sequence of less than 130 base pairs, contained in the large intron of act-4, that appears to function as part of the signal for sex determination. We have attempted to correlate the intersexual phenotype with the presence of the injected DNA using single worm dot blots probed with either vector or insert sequences. One prediction is that all intersexes have the injected DNA while their normal siblings lack it. This proves not to be the case since some of the wild type males have the DNA and, in fact, some of the intersexes do not. This procedure, however, only allows us to detect relatively high copy number sequences (>20,000 copies per worm based on hybridization with Tc1) at the level of the whole adult worm. It is likely that the DNA needs to be present only within specific lineages and at specific stages of development to cause transformation. In addition Stinchcomb et al. ( Mol. and Cell Biol. 5: 3484-3496, 1985) have observed that DNA microinjected into the distal arm of the gonad is maintained as an extra chromosomal array and is subject to mitotic loss. Considering these complications, it is perhaps not surprising that we cannot find a correlation between transformation and detectable levels of the injected sequences. We have tested normal males arising from the injection of pCeA4.4 and found the injected DNA. Thus, this clone is present among the progeny at approximately the same level as the clones that give intersexes. We have used the same assay to examine other X-linked genes for effects on sex determination. A plasmid containing the myosin light chain genes, mlc-1 and mlc-2, (kindly provided by Claudia Cummins and Phil Anderson) has the same hermaphroditizing effect as the act-4 plasmids. This was also true for a phage containing part of the X- linked myosin heavy chain gene, myo-2. A plasmid, pGBA2.2 (provided by Guy Benian and Bob Waterston), containing a 2.2 kb region from the coding region of the same gene, however, had no effect. This means that for all three regions of the X-chromosome tested, at least some sequence has had an effect on sex determination. This leads us to believe that similar sites are very common on the X-chromosome, perhaps as frequent as one per gene. We are currently subcloning both the mlc and myo-2 clones in hopes of identifying a consensus sequence for the X-linked sites involved in the sex determination signal. So far we have injected only one autosomal gene; we are starting injections using other autosomal genes hoping to demonstrate that this sequence is rare or absent from the autosomes.