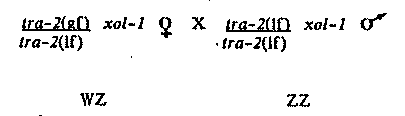Worm Breeder's Gazette 10(2): 18
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
In a previous WBG article (Miller, et al.,October, 1987), we proposed that wildtype xol-1 gene product negatively regulates sdc-1 and sdc-2 to ensure that they, and other genes involved in XX sex determination and dosage compensation, remain inactive in XO animals. XO animals carrying mutations in xol-1 cause the inappropriate expression of sdc-1 and sdc-2, thus allowing the expression of the XX mode of sex determination and dosage compensation. This results in lethality (presumably caused by under expression of X-linked genes) and feminization. In this article, we will provide evidence for reduced X chromosome expression and feminization. We postulated that the XO-specific lethality caused by xol-1 mutations is due to inappropriately low X chromosome expression. To test this, we looked at X-expression directly in dying embryos using the Northern hybridization assay (Meyer and Casson, Cell 47, 871-881) to measure X-specific mRNA levels. We found that xol-1 XO animals under-express their X-linked genes. Using two different X-linked probes (myo-2 and uvt-4), we showed that, relative to autosomal controls (myo-1 and act-1, respectively), the X-linked transcripts were significantly under-expressed in RNA prepared from dying xol-1 embryos when compared to RNA prepared from normal embryos. Quantitation by densitometer tracing of many experiments (over 20) revealed that xol-1 dying embryos express their X-linked genes at levels less than half that of wild-type levels, supporting the theory that mutant XO animals are carrying out the XX mode of dosage compensation. Sex determination in xol-1 XO animals is also shifted to the XX mode. Although xol-1 XO animals die as embryos or young larvae, some of the larvae are healthy enough to allow observation of cells that are sexually dimorphic at hatching. To ensure that we were looking at XO dying larvae, the following cross was made: dpy-21 V; flu-2 were mated with him-8 IV; unc-3 X hermaphrodites. The dpy-21 mutation was used to rescue the xol-1 XO males; the X-linked flu-2 mutation (an intestinal autofluorescence marker scoreable in L1 larvae) was used to identify XO animals. Flu cross progeny from the above cross must be xol-1 XO dying larvae. The sexual phenotypes of three sexually dimorphic cells (B, Y, and HSN) were scored in these larvae and all three cells were found to be capable of undergoing hermaphrodite specific cell fates. Unfortunately, it was very difficult to score the majority of the animals due to the general sickness and extensive cellular disorganization in these animals. As a further complication, the HSN was often impossible to see because the region around the gonad in these dying animals is very refractile. Of 15 scoreable B cells (out of over 50 animals), 11 had chosen the hermaphrodite fate and 4 the male fate. All 7 scoreable Y cells had undergone the hermaphrodite fate. HSNs (hermaphrodite fate) were observed in 3 animals. It is apparent that some cells in xol-1 mutant XO animals undergo hermaphrodite-specific developmental fates, implying that the wild type xol-1 gene product is required in XO animals for proper male sexual development. However, due to the difficulty in scoring most of the animals, it is impossible to estimate the extent to which dying XO animals are transformed. XO animals carrying mutations in xol-1 inappropriately execute the XX mode of both sex determination and dosage compensation. Mutations in genes required for XX sex determination and dosage compensation ( sdc-1 and sdc-2) completely suppress all the defects associated with xol-1 mutations. Doubly mutant animals are essentially 100% viable and all are males. In contrast, mutations in genes required only for dosage compensation (dpy-21, dpy-27, and dpy-28) suppress only the dosage compensation defects (XO lethality and under- expression) but not the sex determination defects (feminization). Doubly mutant animals are alive, but many are still feminized. A typical experiment demonstrating the above points involved mating dpy-21(y47) V; xol-1 X males with rol-6 II; dpy-21(y47) V; xol-1(y9) hrodites. A comparison of Dpy non-Rol non- Unc (XX) cross progeny with Unc non-Dpy non-Rol (XO) cross progeny yields information about the extent of XO rescue. 100% rescue would give equal numbers of XX and XO cross progeny. For this cross, we saw essentially complete rescue (97%). In the case of the temperature- sensitive, maternal effect mutation, dpy-28(y1), we observed (at 20 C) 26% rescue by homozygous dpy-28(y1) progeny from heterozygous mothers and over 80% rescue by dpy-28(y1) homozygous progeny from homozygous mothers. The sexual phenotypes of the XO cross progeny can also be observed and used to show that, with dpy-21, dpy- 27, and dpy-28, many rescued XO animals are still feminized. For example, of the XO cross progeny (Unc non-Dpy non-Rol) in the dpy-21 cross above, 35% were hermaphrodites, 6% were pseudomales, and 59% were males. For XO animals rescued by mutations in dpy-26, ved a higher percentage of hermaphrodites (60%-100%) than with the dpy-21 mutations. When males were mated with XO xol-1 hermaphrodites in the above crosses, two classes of XO cross progeny were observed: one class in which the XO animals carried an X chromosome from the mother and one in which the X chromosome came from the father. Interestingly, the sexual phenotype of a rescued XO animal is dependent upon the parent from which it inherited its X chromosome. XO animals that received a maternal X chromosome were more likely to develop as hermaphrodites, while those that received a paternal X chromosome tended to develop as males. For example, 62% of the dpy-28(y1); rescued XO animals that received a maternal X developed as hermaphrodites, while only 16% of those that inherited a paternal X developed as hermaphrodites. As a final note, it is possible to use a xol-1 mutation to design a WZ/ZZ system of sex determination in C. elegans (similar to J. Hodgkin's system with tra-1). As we have reported before, xol-1 mutations have a masculinizing effect on XX animals that are already partially masculinized by a mutation in the sex determination pathway. For example, a tra-2; X animal can be a mating male. Because mutations in xol-1 allow tra-2(lf) XX males to mate, the genotypes shown below depict a system whereby sex is determined by the activity state of one gene. [See Figure 1]