Worm Breeder's Gazette 10(2): 15
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
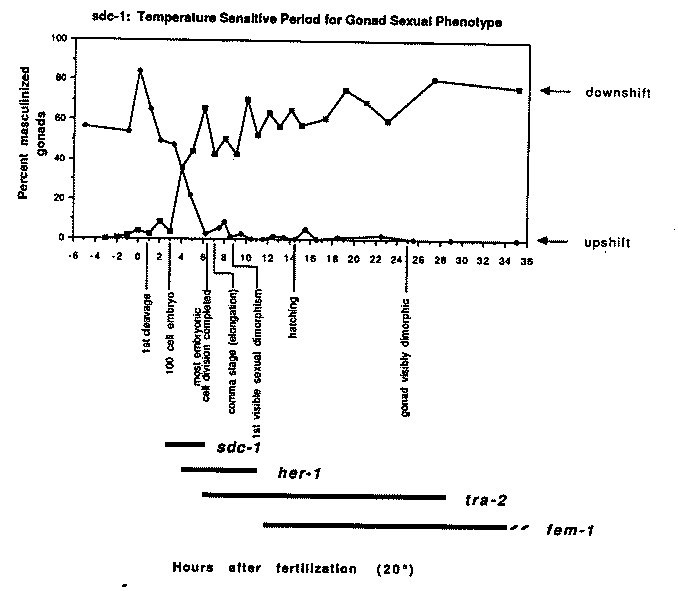
We have been performing temperature-shift experiments using the temperature-sensitive allele sdc-1 (y67) in an attempt to determine the time(s) at which sdc-1 acts in the sex determination and dosage compensation processes. sdc-1 (y67) is sharply temperature-dependent, producing a strong mutant phenotype at 20 C (restrictive) and a nearly wild-type phenotype at 15 C (permissive). Temperature-sensitive period for somatic sexual phenotype: In order to study specifically the effect of temperature on sex determination in the absence of the dosage compensation defect, we have used the temperature sensitive sdc-1 allele in combination with the suppressor mutation y63 (which appears to suppress the dosage compensation but not the sex determination defects of sdc-1 mutations [AMV and BJM, WBG Volume 10:1]). A reciprocal temperature-shift experiment using this strain defines a brief and specific temperature-sensitive period during the first half of embryogenesis for the action of sdc-1 in somatic sex determination; the data for gonad sexual phenotype are depicted in the accompanying graph. The tsps for both somatic gonad and tail/copulatory structures are coincident, extending from approximately the 100-cell stage to the end of the majority of embryonic cell division (approximately 320 minutes after the first cleavage), just prior to the onset of elongation. This period is complete more than two hours before the first visible sexual dimorphism (HSN cell death in the male), approximately eight hours before hatching, and nearly a full day before the gonad itself becomes sexually dimorphic. We have previously proposed that sdc-1 acts upstream of her-1 in the sex determination pathway, as a negative regulator of her-1 activity in XX animals. Consistent with this proposal, the tsp for gonad sexual phenotype for sdc-1 is prior to that determined for her-1 (J. Hodgkin, JEEM 83, Supplement, 103-117). As with any t-shift experiment, we cannot rule out the possibility that the tsp might reflect the time of synthesis of a thermostable product rather than the time of action of a thermolabile product. The fact that the upshift curve has an inflection point during embryogenesis suggests that at least the maternal endowment of sdc-1 ( y67) may be thermolabile, since sdc-1 mutants are normally rescued by maternal sdc-1+ activity. This is not a strong argument, however, since 1) 15 C sdc-1(y67) product may not be equivalent to wild-type sdc-1 for maternal rescue activity and/or 2) the maternal endowment of sdc-1 may be supplied as RNA. Effects of temperature on the sdc-1 dosage compensation phenotypes: We have begun to assess the effects of temperature on the dosage compensation phenotypes of sdc-1(y67) . This analysis is somewhat less straightforward than that for sexual phenotype, since the visible morphological manifestations of the dosage compensation defect (Sma, Egl, protruding vulva) are more ambiguous to score than is the case for sexual transformation. Moreover, increased X-expression only late in development may not have any obvious scorable morphological consequences. As a first step, we have performed reciprocal shift experiments defining the tsp for the 'Sma enotype using a her-1; ) strain. The upshift curve in this case closely resembles that for somatic sexual phenotype; that is, remaining at permissive temperature through the end of embryonic cell proliferation is sufficient to yield a nearly wild-type phenotype. The simplest interpretation of these data is that the presence of sdc-1 activity through the first half of embryogenesis is sufficient for establishment of the normal XX mode of dosage compensation. The downshift curve, however, is quite different- - shifting from restrictive to permissive temperature even as late as L2 can result in partial rescue of the mutant phenotype. It seems that 'wild-type' sdc-1 activity may be capable of shifting animals back toward the XX mode of dosage compensation even when introduced at a time later than sdc-1 normally acts during wild-type development. Conclusions about the function of sdc-1 in dosage compensation based on these experiments must of course remain tentative pending demonstration that these morphological phenotypes accurately reflect the state of X-linked gene expression. We are currently assaying levels X-specific transcripts in RNA preparations from synchronized populations of sdc-1(y67) worms that have been raised at one temperature through embryogenesis and then subsequently shifted to the second temperature for the remainder of development. Tail and gonad do not choose their sexual fates independently in sdc- 1 mutants: A striking observation has come out of careful analysis of the classes of sexually transformed animals produced by sdc-1 mutant strains in which animals range in sexual phenotype from fertile hermaphrodites to intersexes to pseudomales. Specifically, there is a strong correlation between tail and gonad sexual phenotypes in individual animals even though these tissues diverge lineally at the first embryonic cleavage. XX animals with male tails usually have male gonads, and animals with hermaphrodite tails usually have hermaphrodite gonads, while animals with tail and gonad of opposite sex are relatively rare. Put more concisely, different cells in the embryo are not choosing their sexual fates independently. How can we explain this phenomenon mechanistically? One possibility is that there is a strong influence of the maternally provided environment on the choice of sexual fate. The complement of components supplied to the oocyte by the mother is likely to vary somewhat from oocyte to oocyte; in the absence of sdc-1 activity, the choice of sexual fate becomes sensitized to this naturally-occurring variability. (This phenomenon probably cannot be accounted for simply by residual maternal sdc-1 activity resulting from mutations that do not entirely eliminate gene function, since it occurs with all sdc-1 alleles including those likely to represent or approximate the null phenotype.) A second intriguing possibility is that the sex determination decision in C. elegans is not cell autonomous, but rather involves cell-cell interactions. This type of model suggests the existence of a 'master' cell or tissue in which the X/A ratio is assessed and the decision is made; that tissue or cell then communicates with the rest of the cells in the worm, perhaps via humoral factors, to regulate downstream switch genes such as tra-1 that control sexual identity in a cell-autonomous fashion (C. Hunter and W. Wood, WBG Volume 10:1). We are now preparing to do mosaic analysis of sdc-1 with the hope of shedding light on these issues.