Worm Breeder's Gazette 10(2): 118
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
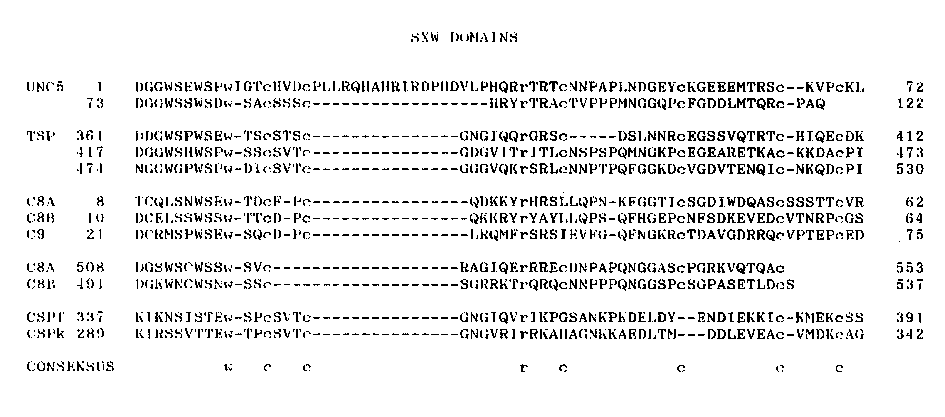
In the last gazette we reported the cloning of a 0.6 KB EcoRI fragment from the unc-5 region which appeared to have a Tc1 insertion responsible for the unc-5(ev435:Tc1) mutation. We have identified several clones in an EMBL3 library of N2 DNA which hybridize to this fragment. Most clones appear to have part of the 0.6 KB EcoRI fragment attached to one of the lambda arms. Clones NW#11, NW#12 and NW#13, however, contain the entire 0.6 KB EcoRI fragment, which appears by sequence analysis to be identical to the 0.6 KB EcoRI fragment used as probe. NW#11 and Nw#13 appear identical to each other, whereas, clone NW#12 differs from these only slightly. Southern blots of EcoRI digested wild type DNA revealed approximately 23 EcoRI fragments which hybridized to NW#11 DNA. Polymorphisms in several of the unc-5 mutations (ev435, ev440, ev442, h571, rh1036, rh1037, and rh1038) isolated in RW7097 were revealed using NW#11 DNA to probe Southerns. Sequence analysis of the 0.6 KB EcoRI genomic fragment revealed a relatively GC rich putative open reading frame of 122 amino acids flanked at 5' and 3' ends by consensus splice acceptor and splice donor sites and AT rich DNA. The Tc1 insertion site of unc- 5(ev435:Tc1) is near the middle of this open reading frame. We have also isolated a 1 KB cDNA by hybridization to the 0.6 KB EcoRI fragment. A portion of the sequence of this cDNA is identical to that of the genomic fragment, beginning at the consensus splice acceptor site and continuing into the open reading frame. The hypothetical protein fragment of 122 residues encoded by the unc-5 exon comprises a tandemly arranged pair of homologous domains each beginning with the tryptophan-rich sequence DGGWSXWSXW. Three similar domains are present in human thrombospondin where they are also tandemly arranged ( 1). Thrombospondin (TSP) is a large glycoprotein secreted by blood platelets and other cells, which promotes cell aggregation and cell- substratum adhesion in vitro. The region of thrombospondin that contains these domains is believed to bind to various extracellular matrix components including type V collagen, laminin, fibronectin, fibrinogen, and plasminogen. A similar domain occurs twice in complement components C8 alpha and C8 beta, once near the amino terminus and once at the carboxy terminus. Only the amino domain occurs in complement component C9. A single related domain occurs once in the circumsporozite (CS) proteins of Plasmodium falciparum and Plasmodium knowlesi, malarial parasites of humans and Old World monkeys (2,3). It has been suggested that the CS proteins may promote parasite adhesion during liver invasion. The occurrence of this common amino acid sequence in apparently unrelated proteins involved in cell adhesion suggests that it may fold and function as an independent protein domain especially adapted for binding to extracellular matrix components. It may be interesting to learn whether each such domain in thrombospondin or the hypothetical unc-5 protein binds to a distinct ligand. [See Figure 1]