Worm Breeder's Gazette 10(1): 89
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
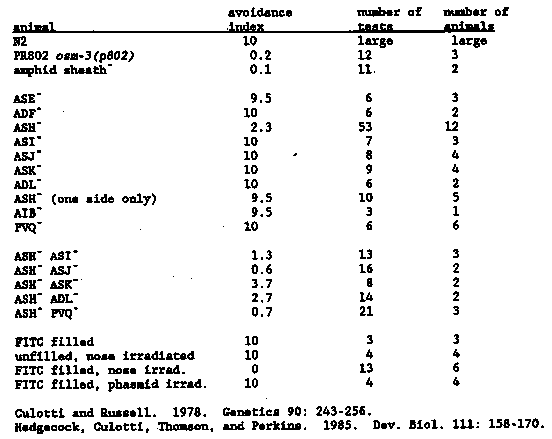
When a worm encounters a region of high osmotic strength with the tip of its nose, it responds by backing up, typically several body lengths, and usually turning around. Genetic analysis of this behavior should be powerful, since selection of animals that fail to respond to osmotic strength is simple. To determine which sensory neurons mediate the osmotic avoidance response (hereafter called the Osm phenotype), we used a laser to kill neurons in early L1 larvae, and performed single-animal Osm assays on the animals as adults. For a discussion of various concerns about the validity of laser-killing experiments on neurons, see the accompanying newsletter entry (Thomas, Garriga, Avery, and Horvitz). Our assay is a modification of that used by Culotti and Russell ( 1978). About 15 l of 4 M fructose (plus Congo Red, a dye that worms do not respond to) is applied to a dry NG plate in a ring with an inner diameter of about 0.8 cm and allowed to soak into the agar. A single animal is moved without bacteria onto the test plate inside the osmotic ring and allowed to run for 15 minutes, with visual checks at least once per minute (tracks are observed at the end of the assay to be sure that a transient crossing did not occur). The time the animal takes to cross the osmotic barrier is recorded, rounded to the nearest minute. The minimum score is one minute. Notes on individual encounters are usually made as well. A wild-type animal usually encounters the fructose ring in fewer than 30 seconds, responds normally, and heads off in a new direction. In a typical assay the animal will encounter the ring about 20 times in 15 minutes. Wild- type animals very rarely cross the ring, and then only very late in the assay (when the osmotic barrier has diffused considerably). When a mutant such as osm-3(p802) is run in this assay, the animal typically crosses the barrier in less than one minute, without any response [See Figure 1]. From the results of several such tests we calculate an osmotic avoidance index (OAI) - 10/14 (t - 1), where t is the mean time to cross. This produces a range of OAI from 0 to 10. The OAI for N2 is very close to 10 and that of osm-3(p802) is 0.2 (osm- 3 animals never respond to the osmotic barrier, but sometimes take longer than one minute to cross). What sensory neurons mediate this behavior? We knew the sensing was likely to be in the head, and the only known head defect of osm-3 animals is ultrastructural abnormalities in the amphid sensory endings. Therefore we killed the amphid sheath cell (both sides) to test whether the Osm response was eliminated. It was (see Table for a summary of all of the cell killing data). Since the sheath acts as a glial-like sheet around all eight of the amphid chemosensory endings, it seemed likely that one or more of these neurons was mediating Osm. We FITC-filled six of the amphid chemosensory neurons (ADF, ASH, ASI, ASJ, ASK, ADL), by the method of Hedgecock et al. (1985), and bleached the dye using a fluorescein filter set and an epifluorescence light source. This treatment also eliminated the Osm response (see Table for data and controls). With the laser we killed seven of the eight amphid chemosensory neuron types individually and tested each for osmotic avoidance (the eighth neuron, ASG, has not been done because it is more difficult to identify with certainty and is not FITC-filled.) In addition, the amphid interneurons AIB and PVQ were killed. Only when ASH was killed were animals significantly less responsive than normal. ASH animals frequently failed to respond to the osmotic barrier at all, but sometimes responded and crossed only later in the assay. Even when an ASH animal responded it was usually deeper in the osmotic barrier than normal, suggesting a less sensitive or slower response. Killing ASH reduces the efficacy of the Osm response, but it is not the whole story. Any other sensory neuron involved in the Osm response should also be in the amphid, and probably one of the FITC-filled cells, because of the amphid sheath and FITC-filling results. We searched for a second neuron by doing double kills with ASH (both sides) and a second amphid sensory neuron (both sides). Of the four neurons tested, only killing ASJ effectively eliminated the residual Osm response (we are not yet sure whether the apparent reduction by killing ASI is significant; we have not yet tested ADF). ASJ has output only to PVQ, an interneuron. Killing PVQ and ASH also eliminated the residual Osm sensitivity, suggesting that ASJ acts through PVQ. We should emphasize that the assignment of ASJ and PVQ to the Osm response is tentative; we must kill these cells together with ASH in more animals to be certain of this relatively marginal effect. We anticipate that further application of the laser to the Osm behavior will clarify the rest of the neuronal pathway from sensory input to motor response. Identification of the cells in this pathway will assist in the interpretation of mutants defective in the Osm response.