Worm Breeder's Gazette 10(1): 79
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
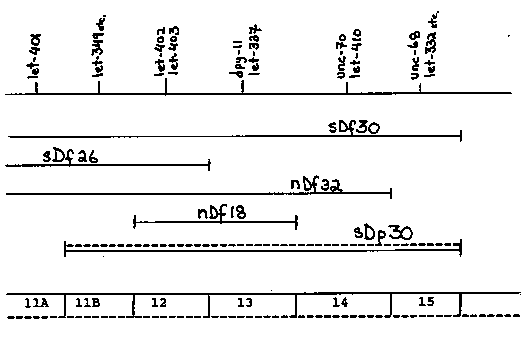
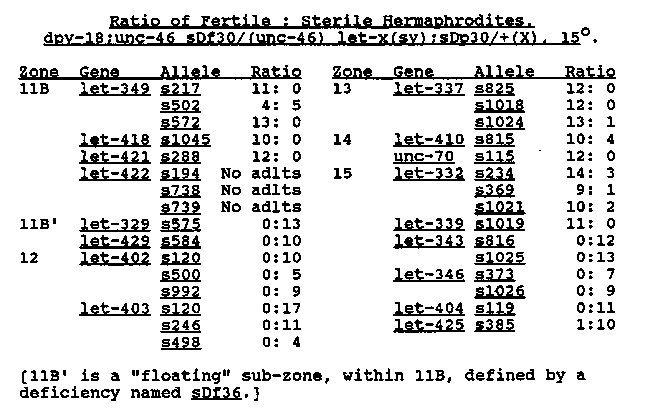
The duplication sDp30(V:X), on the X chromosome, carries wild type copies of dpy-11(V) and surrounding LGV genes. It arose in a strain carrying a gamma ray induced deficiency, sDf30(V), and presumably was a transposition of most (but not all) the genes deleted by the deficiency. [See Figure 1] The duplication was discovered when conflicting results were obtained from reciprocal complementation tests between the following individuals: dpy-18/dpy-18(III):unc-46 crossed to dpy-18/dpy-18(III);(unc-46) /eT1(V), - [where let-x(sy) was an allele of one of the genes in zones 11B-15 and, in some cases, was linked to unc-46]. Adult Dpy were produced when sDf30 and sDP30 were carried by the hermaphrodite parent, but no such males were produced from the reciprocal crosses. Suppression of the lethal phenotypes by the duplication was not uniform in F1 hermaphrodites having the genotype dpy-18:unc-46 sDf30/( unc-46) let-x(sv);sDp30/+(X). At 15 C most of these hermaphrodites reached adulthood, but not all were fertile (Table), even though the stock strains, maintained at 15 C with the same let gene dosage [dpy- 18/eT1:(unc-46) let-x/eT1], were fertile. This suggests that some genes were under-expressed in the duplication. [See Figure 2] The under-expression of some of the genes may be related to the phenomenon of X-dosage compensation, known to occur in C. elegans ( Meneely & Wood, Genetics 117: 25,1987), which involves the equalizing of X-linked mRNA transcripts in the two sexes (Meyer and Casson, Cell 47: 871,1986). Whether compensation is achieved by elevating expression in the single X chromosomes of males or by reducing expression in the two X chromosomes of hermaphrodites or by a combination of these two mechanisms is not yet clear. Based on data of Meneely & Wood (1987), using dpy-21 and dpy-26, and those of Meyer & Casson (1986), using dpy-27 and dpy-28, reduction of X chromosome expression in hermaphrodites appears to be part of the mechanism. We may, therefore, speculate that in our experiments (1) reduction of X chromosome expression occurred in hermaphrodites, (2) this reduction spread to the linked autosomal genes on sDP30 and (3) either the genes were variably affected by the spreading effect or the mutant phenotypes were variably sensitive to reduced doses of wild type product. To test whether the apparent under-expression of certain sDp30 genes was due to the effects of X-dosage compensation, we plan to study sDp30's properties in the background of dpy-21 which is known to effect dosage compensation (Meneely & Wood 1987). In addition, we hope to acquire new duplications that carry the same genes as sDP30 but are transposed to an autosome. Supported by grants from NSERC and MDA of Canada to DLB.