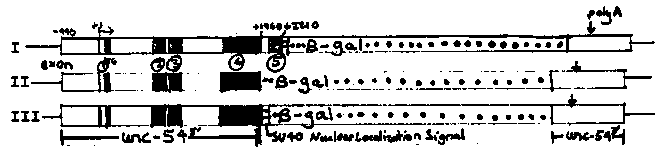Worm Breeder's Gazette 10(1): 60
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
As reported in a previous gazette article (A. F. and R. Waterston WBG #9.3), cosmids carrying the unc-54 gene contain all of the cis- acting information necessary for proper expression of the gene. We are attempting to define the sequence elements mediating body wall specific gene expression. To do so, we constructed plasmids carrying unc-54 sequence which have been deleted and mutated in vitro. These plasmids are injected into oocytes of animals carrying the mutation unc-54(e190). e190 is a deletion eliminating unc-54 product [the major myosin heavy chain protein] and greatly diminishing unc-54 message levels. This allows both transient and stable expression assays for the injected DNA. Transient Expression: After injection of functional unc-54 plasmids, a high fraction (5%-25%) of the injected oocytes give rise to rescued progeny, as assayed both by improved movement and egg laying. Stable Transformation: Most of the transiently rescued animals do not breed true; that is all progeny are unc. In a small fraction of cases the rescued animal has some rescued progeny and a transformed line is established. Frequencies of stable transformation vary greatly (3-20% of injected animals), with the factors influencing transformation frequency still not understood. Stably transformed lines are grown up and analyzed for the presence of DNA and RNA from the initial plasmid, and stained with monoclonal antibodies (from David Miller) to localize unc-54 protein expression. An anti myo-3 monoclonal with a different fluorescent tag is used as internal control and marker; unc-54 and myo-3 proteins localize to the same muscle cells in wild type animals. A considerable variability in the level of expression from the transgenic loci constructed with the same input DNA is observed. The level of expression does not correlate with the copy number of the injected DNA and is thus likely to reflect modest position or contextual effects. Copy numbers in the lines vary from 0.5-6 per haploid genome. A single exceptional line with about 60 copies of plasmid pUNR-54 (used as a starting point for the deletions) actually expresses very poorly. The unc-54 gene does not require extensive flanking sequences for proper expression. On the 3' side, deletions which leave 1000nt beyond the poly A site exhibit normal expression. More surprisingly, almost no 5' sequence seems to be required for expression. The startpoint of transcription has been mapped to 80-85 nt upstream of the AUG (Originally done by Mike Krause and repeated by us.) Deletions which eliminate some or all of the upstream region still function in both transient expression and stable transformation, with the wild type pattern of expression observed in all of the stably transformed lines. More surprisingly, a deletion which eliminates the entire 5' flanking region and 50 nt of the transcribed leader sequence is also functional in both transient expression and stable transformation. A transformed line carrying this deletion expresses unc-54 myosin in exactly the correct subset of cells. Reassuringly, a deletion which removes the AUG and first four codons is nonfunctional for expression. Primer extension analysis of RNA from strains rescued with 550 or 130 nt of DNA upstream from the transcriptional start show the wild type pattern, suggesting that 130 nt of 5' flanking DNA is sufficient for proper initiation of transcription as well as proper regulation. RNA from the line transformed with the +50 deletion seems to start at a set of sites just adjacent to the gene within plasmid sequences. This suggests that a tissue specific regulatory element (an enhancer?) within the transcribed region can stimulate transcription from a variety of upstream start sites. Expression of unc-54: -galactosidase fusion proteins: Three different unc-54: -gal fusion proteins have been integrated into the genome of tra-3(e1107am) animals using sup-7 as a selectable marker. These fusions were constructed conservatively so that both the 5' and 3' ends of the resulting chimeric gene derive from unc-54. Each of these proteins is expressed at high levels in transformed lines. In each case (four lines total), the expression is limited to the body wall type muscle cells. In the first two cases the staining is throughout the cells, with distinct blue staining outlining the four muscle quadrants. In the third case the staining is primarily nuclear with each of the body wall type muscle nuclei staining dark blue. Expression of these fusions is much higher (10-100 fold) than the expression of the HSP: -gal and myo-2: -gal fusions that has been reported previously. Whether this results from differences in transcription due to the unc-54 'promoter' or to differences in message stability due to 5' and 3' unc-54 RNA sequences is not yet known. The ability of the SV40 nuclear localization signal to function in construct III but not construct I presumably reflects different contexts of that region within the two proteins upon folding. Taken together, the whole gene expression data and the -gal fusion data suggest that a determinant for tissue specificity is located in one of two regions (+50 to +1960 from the start of transcription or - 240 to +970 from the polyadenylation site).