Worm Breeder's Gazette 10(1): 131
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
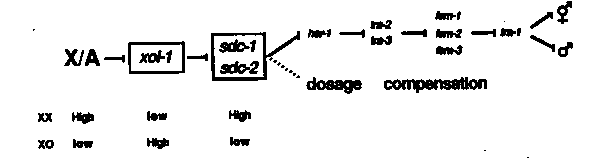
xol-1 is required in XO animals for proper sex determination and dosage compensation. Mutations in the gene xol-1 result in XO-specific lethality and map between mec-7 and dpy-6 on the X chromosome. The lethal phenotype is recessive since stDp2/f; xol-1 XO mutant animals are viable males. Two independently isolated mutations, y9 and y70, both exhibit XO- specific lethality and map to the same interval on X. In addition, they are thus far inseparable by recombination (<.01 m.u. apart), indicating that they are probably alleles of the same gene. The time of death for xol-1 XO mutant animals extends from mid- embryogenesis to early in the L1 larval stage. It is likely that these XO animals die because, in the absence of xol-1, the XX mode of dosage compensation is being expressed in XO animals, presumably resulting in under expression of X-linked genes. The XO lethality of the xol-1 mutations can be rescued by mutations in dpy-21, dpy-27, and dpy-28, which themselves elevate X- linked gene expression in XX animals. Presumably, mutations in these dpy genes prevent proper expression of the hermaphrodite mode of dosage compensation that is being inappropriately expressed in xol-1 mutant XO animals and thereby return levels of X-linked gene expression to normal. Consistent with this idea is the finding that there are wild-type levels of X-linked mRNA in dpy-28(y1); and dpy-21(e428); XO animals. The XO animals rescued by mutations in dpy-21, 26, 27, and 28 range in phenotype from fertile males to fertile hermaphrodites. Therefore, although both y9 and y70 were isolated as apparent 'suppressors' of the dpy-28(y1) mutation, though non-Dpy (unsuppressed) animals in the y1; y9 and y1; y70 strains are, in fact, XO animals that have been transformed into hermaphrodites by the xol-1 mutations and are not suppressed XX animals. In a typical experiment that demonstrates transformation of XO animals into hermaphrodites, y1; y70 males were mated with dpy-18(e364) ); ) 1) hermaphrodites. Among the cross progeny were many Unc nonDpy (XO) hermaphrodites. Similar results have been obtained with mutations in dpy-21 and dpy-27. Although mutations in dpy-26 have not yet been tested as directly, results from other experiments indicate that many dpy-26; t animals are also transformed into hermaphrodites. The transformation of XO animals into hermaphrodites in the above strains suggested a role for xol-1 in sex determination and prompted us to look directly at the dying embryos and L1 larvae in xol-1(y9) and xol-1(y70) mutant strains. It is possible to determine the sex of dying L1 larvae in these strains by observing cells that are sexually dimorphic at hatching. Preliminary observations suggest that B, Y, and HSN cells can undergo hermaphrodite-specific cell fates in dying xol-1 mutant L1 larvae, implying that xol-1 is required in XO animals for proper sexual development. It appears that mutations in dpy-21, 26, 27, and 28 can suppress the dosage compensation defects but not the sex determination defects found in XO xol-1 mutant animals. By contrast, mutations in sdc-1, a gene required for hermaphrodite development and proper X expression in XX animals (Villeneuve and Meyer, Cell 48: 25-37, 1987), are fully epistatic to xol-1 mutations in that they are able to suppress both the sex determination and the dosage compensation defects of xol-1 mutations in XO animals. Specifically, all xol-1(y9) 5) XO animals are viable males (A. Villeneuve, personal comm.). The same result was observed with a newly identified gene, sdc-2 (C. Nusbaum and B. Meyer, this WBG). In a possible model explaining xol-1's function in XO animals, the wild-type xol-1 gene ensures that the activities of sdc-1, r genes involved in hermaphrodite sex determination and dosage compensation remain inactive in XO animals (see figure below). Mutations in xol-1 also affect sex determination in XX animals. Although xol-1 mutant XX animals are phenotypically wild type, xol-1 mutations further masculinize XX animals that are already partially masculinized. tra-2(strong) and tra-3(strong) mutations result in the incomplete transformation of XX animals into non-mating pseudomales. Surprisingly, the xol-1(y9) mutation, in combination with these strong alleles or n1106 (a weak allele of tra-2) causes further transformation of some XX animals into mating males. y9 also dramatically masculinizes her-1(n695sd) XX animals. In a her-1(n695sd) strain, 30% of the XX animals are nonmating pseudomales while the remaining 70% are Egl hermaphrodites (C. Trent, personal comm.). In a her-1(n695sd); strain, all of the XX animals are at least partially transformed into pseudomales and some are capable of mating. Furthermore, y9 enhances the Tra phenotype of sdc-1 XX animals, although mating males are not produced. xol-1(y70) also displays an XX phenotype similar to y9 in that it fails to complement y9 in the further masculinization of some tra-2( strong) XX animals into mating males. y70, however, appears to be much weaker than y9 with respect to this XX sexual transformation phenotype. A loss-of-function mutation in her-1 does not block xol-1(y9)'s masculinization of a strong tra-2 mutation, implying that wild-type her-1 product is not required for further masculinization. Furthermore, mutations in fem-1, and strong mutations in tra-1 are all epistatic to the xol-1(y9) mutation. Therefore, with respect to maculinization of XX animals, xol-1 either interacts with the major sex determination pathway somewhere downstream of her-1 and upstream of tra-1 or interacts in a parallel pathway. A small, X-linked deficiency that spans the xol-1 locus, uDf1 ( provided by the Chalfie lab), fails to complement the XX sex determination phenotype of y9. Specifically, some tra-2(strong); y9/uDf1 XX animals are mating males. Preliminary experiments with uDf1 have revealed that y9 and y70 are probably loss of function mutations in xol-1 and that the xol-1 locus is haplo-insufficient with respect to its interactions with her-1(n695). Some XX animals of both genotypes her-1(n695); y9/f and her-1(n695); uDf1/+ (D. Hsu and B. Meyer, this WBG) are mating males. Moreover, some XX animals of the triply heterozygous genotype tra-2(strong)/+; 5)/+; y9/+ are mating males (D. Hsu, personal comm.). In addition, neither y9 nor y70 is suppressed by the tRNA amber suppressor sup-5(e1464) in either one or two copies. Reversion of xol-1(y9 or y70) has been undertaken by many members of our lab and has yielded many new alleles of previously identified genes. Using the xol-1 reversion screen, we have isolated 13 alleles of dpy-21 (one of which was induced in a mutator strain provided by the Anderson lab), 1 allele of dpy-26, 5 alleles of dpy-27, and 2 alleles of sdc-1. 7 additional alleles of tra-1 and 17 alleles of tra- 2 were also isolated. In addition, the xol-1 reversion screen has allowed the isolation of a new Tra mutation on V and another one on X. Furthermore, 21 alleles of sdc-2 have been isolated as suppressors of y9 (one in a mutator background).