CelegansChrome: a Google Chrome Browser Extension for C. elegans Research
2Institute for Neuroscience, George Washington University
Browser extensions are small software programs that enhance the user’s online experience by creating greater browser functionality. Here, I present a Caenorhabditis elegans specific extension for Google Chrome browsers. The software is built from four files: 1) an HTML file called celegansChrome.html that generates the user interface; 2) a JavaScript file called celegansChrome.js that contains objects of C. elegans gene data; 3) a JSON file that contains a manifest of the program contents called manifest.json; and 4) a small image file of a C. elegans hermaphrodite called icon.png that gets loaded into the browser toolbar. The software is freely available at the Google Chrome Web Store by simply clicking the add to Chrome link to load the extension into the browser:
chrome.google.com/webstore/detail/celeganschrome/ipnlfcanpnhkhljmminidjndijjmheib
The user can enter a gene common name (e.g., egl-4) and the Chrome extension can provide the user with C. elegans gene data from any website or even while off-line. Often, it’s convenient to retrieve linkage data or interactome data for a specific C. elegans gene while browsing different webpages or reading journal articles online – this extension provides a frictionless interface for the user to retrieve these data without having to open a new tab and search another website or paper for these data. Currently, the extension provides the user with linkage data for C. elegans genes, gene interaction data, human othology data to a C. elegans gene, and also a gene overview using the WormBase (Harris et al., 2014) RESTful API (only this feature requires internet connectivity). The linkage data loaded into the C. elegans Chrome extension was mined from WormMart WS220 (www.wormbase.org/), while the human ortholog data is from the InParanoid program database (Sonnhammer and Ostlund. 2015). The interaction data were downloaded from the WormBase version WS237 ftp site, and the overview of gene data relies on the WormBase RESTful API. The WormBase RESTful API provides access to gene data by creating a URI using standard HTTP requests that is unique to each gene – the unique identifier being a WBID. The C. elegans Chrome extension takes as input the C. elegans gene common name and maps it to the corresponding WBID to generate the unique URI.
This simple browser extension operates as a client-side program (with the exception of the overview option), and thus provides very fast and seamless data retrieval for C. elegans gene data while browsing any site online.
Worm Humor
Web searches conducted by male lab members reveal new postdoctoral candidate to be attractive, possibly single. (Boston, MA). An Internet search encompassing laboratory websites, Facebook accounts, and an intramural volleyball team webpage has revealed Andrea Brooks, a soon-to-be-interviewed postdoctoral candidate, as attractive and possibly unattached. The thorough study was conducted by a team of male postdocs and graduate students within the lab in an effort to “properly vet” the candidate prior to her upcoming interview, sources reported. Although poor annotation of group lab pictures led to some initial uncertainty regarding the identity of the candidate, additional research unambiguously identified Brooks (third from left) as a statuesque brunette with shoulder-length straight hair. Although social media sites indicate Brooks to be single, the presence of an athletic male seated next to Brooks at a lab function and behind Brooks in a Beta-Blockers team picture has led to some speculation that Brooks may be attached. Although any discussion regarding the potential for future romantic involvement with the candidate was carefully avoided by lab members, third-year postdoc Kevin Pollicott stated that while he could see good reasons to keep work and personal life separate, “Sometimes you just have to let things happen”. “Obviously, it would be different if she was an undergraduate,” stated Pollicott emphatically, “unless she was like super mature or something”. Notably, Brooks’ pending interview has generated the highest level of personnel interest since 2012 when first-year graduate students Lisa Sundaram and Jennifer Chu simultaneously carried out rotations. Talks are also underway to start a lab volleyball team.
Nation’s PIs don’t know why your PCR didn’t work. (Bethesda, MD). In a press conference held on the campus of the National Institutes of Health, the Nation’s Principal Investigators (PIs) issued a joint statement to underscore that they have “no clue” why your PCR reactions didn’t work this time. “Really, we have no idea whatsoever – have you tried changing all your reagents, or at least the nucleotides?” added the Nation’s PIs. In addition, the Nation’s PIs emphasized that they will also be unlikely to provide any insight as to why your PCR reactions will undoubtedly fail in the future. “Have you BLAST’d your primers, run a positive control, or tried a gradient?” questioned the PIs. Private sources also indicated that the Nation’s PIs were additionally concerned that you failed to add all the reagents or possibly even used that old Taq in the back of the frost-free freezer that everyone knows is garbage. “What exactly do you want me to tell you?” questioned the Nation’s PIs rhetorically, “How am I supposed to have any inkling as to why you have a giant smear down the entire length of the gel? Did you even run any markers?” An under-the-breath muttered response from the Nation’s Graduate Students is expected to be issued in the near future, which may address the fact that the Nation’s PIs “don’t even know how to program the PCR machines in their own labs” and have to ask for help every time they decide to waste a couple microliters of Phusion on some half-baked experiment that will be “abandoned before the 21st cycle is even completed”.
Questions rejected by the CRISPR Panel at the 2015 International C. elegans Meeting.
Dear CRISPR Panel,
My wife and I attend regular potluck dinners with a number of couples that we’ve known for many years. We like everyone’s company and these are enjoyable social gatherings. The problem is that while my wife and I put a lot of effort into making creative and nutritious meals, some of the other couples think it’s OK to just bring leftovers or salad out of a bag. I don’t want to be a wet towel or to alienate other guests, but it seems as though everyone should be putting equal effort into the meals we are sharing. Should I say something?
Also, my CRISPR isn’t working.
Sincerely,
Marvin Abelstein
——————————————-
Dear CRISPR Panel,
I am having trouble with the women in my lab crying every time CRISPR doesn’t work. Any suggestions would be greatly appreciated!
Sincerely,
T.H.
——————————————
My Dear Beloved:
I know this message will come as surprise but it is great honorific that from displaced murdered royal family I ask assistance in this the transfer of US$2.4M from Bank in Cotonou Benin Republic West Africa.
Please I will offer your trustworthy person 20% of total sum and ask that you reply urgently so that I may return to my medical studies.
Awaiting your generous reply and also having trouble with CRISPR, especially the incorporation of large inserts.
Your Faithful Servant,
Nwangu Akpabio
Preprints and open archives: an open letter to the community from Marty Chalfie
I have just returned from a very exciting meeting on archiving of manuscripts (preprints) in the biological sciences organized by Daniel Colón Ramos, Jessica Polka, Ron Vale, and Harold Varmus (See asapbio.org for more information), and I have become a believer. I am writing to encourage you to join me in changing the way that biological results are made available to the scientific community by submitting your work to an online archive at the same time or even before you submit it for publication in a traditional journal.
As many of you know manuscripts in physics, mathematics, computer science, nonlinear sciences, quantitative biology and statistics are almost universally submitted to ArXiv.org, so they are available to interested readers when the authors feel they should be made public. Approximately 80% of these manuscripts are also submitted to traditional journals, sometimes with modifications suggested by readers and even new coauthors. Submission to arXiv has become the standard in these fields. Similar archives have been developed for the biological sciences (See asapbio.org/resources). Most journals already permit preprint archiving (the biggest exceptions are the Cell Press journals; See Wikipedia’s list of journal preprint policies and SHERPA/RoMEO).
From the discussions at ASAPbio, I have come away with the following thoughts. First, manuscript submission to an open archive will speed up science. The current system of manuscript submission to journals [with review, editorial evaluation at various stages (and all to often resubmission and review), and embargo] means that work ready for public distribution is delayed for months and even years. Depositing the manuscript in a preprint archive will eliminate this downtime (even more so when the manuscript may have to be submitted to another journal). Moreover, a broader audience will have time to help improve the manuscript before its final publication. Second, the work will be universally available. All scientists and interested readers, especially those not associated with rich institutions or countries, will have equal access to the material.
As such, manuscript archiving is an important step in democratizing scientific information. Third, submission to a manuscript archive establishes a time stamp documenting when results and ideas were first presented to the general scientific community. Not only can such a time stamp demonstrate priority, but because multiple version of the work can be archived, this repository can help future science historians follow the development of ideas and discoveries. Fourth, the general consensus at the meeting was that such manuscripts, even though not peer reviewed, should be considered for hiring, promotion, and grant reviews (although we all have to work on our institutions to enable this change). Such a change not only allows a more inclusive view of a person’s work, but frees these review decisions from the vagaries of the manuscript reviewing process.
What am I going to do? When I returned to the lab, I found an email asking for a revision of a paper we had previously submitted to Mol. Biol. Cell. Today, we submitted our revised manuscript both to the journal and to bioRxiv.org. We will submit our future manuscripts and revisions simultaneously to bioRxiv.org and the journals we wish to publish in. We welcome comments, suggestions, and criticisms of this work as it goes online and hope that the more rapid dissemination of our results will be helpful to others.
What can you do? The C. elegans community has always been known for its collegiality and helpfulness. The Worm Breeders Gazette was established so researchers could share their work with the community and for many years it served to promote collaboration, excitement, and camaraderie. I hope that submission of all our work to bioRxiv.org or another archive will continue this tradition and help all of our research.
All the best,
Marty Chalfie
Drafted: February 19th, 2016
Posted to the WormBook News mailing list: March 10th, 2016
Rapid marking of hard-to-score mutations by CRISPR/Cas9-mediated generation of closely-linked, easily-visible mutations
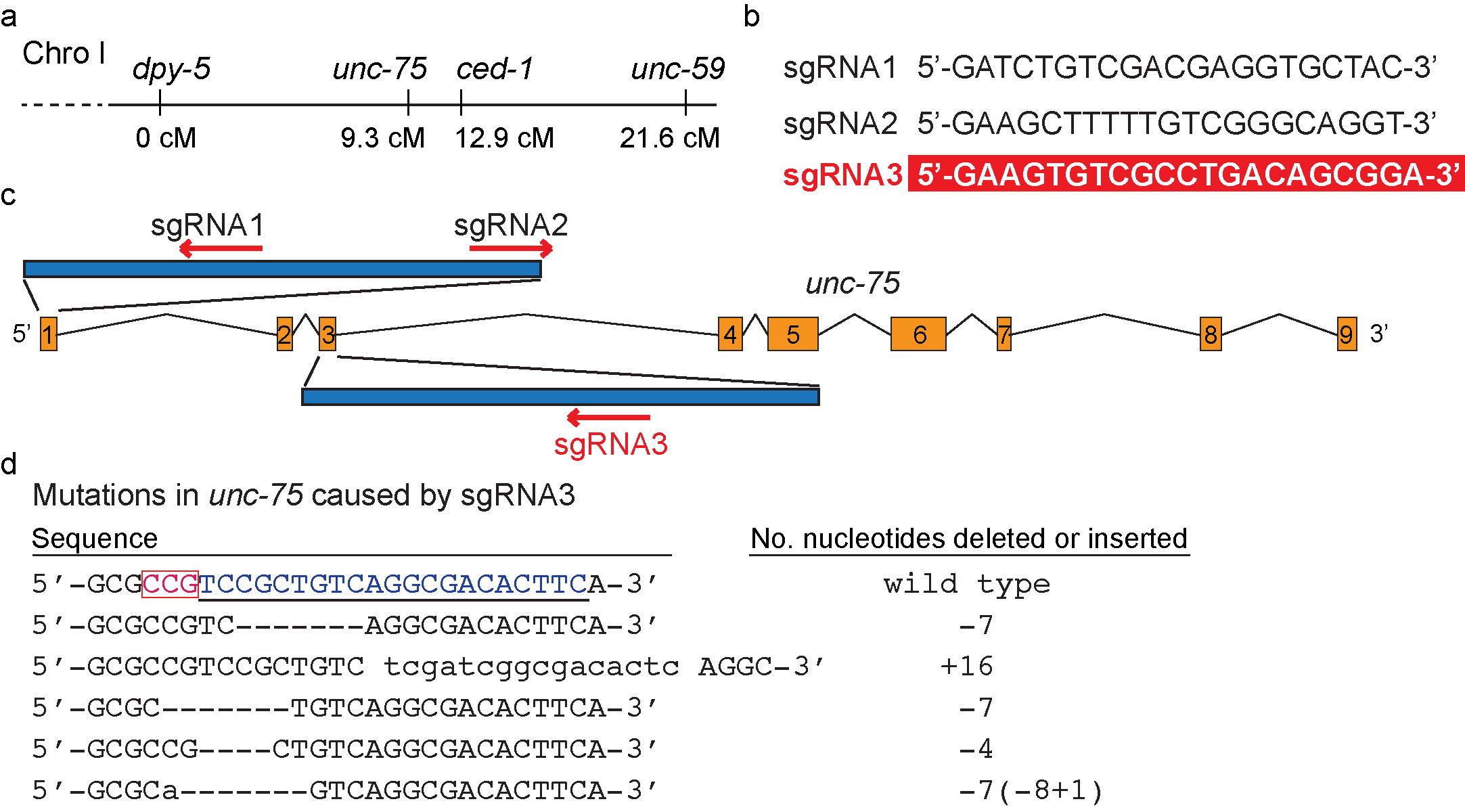
Genetic analysis of mutations that result in phenotypes that are relatively hard to score (weak or partially penetrant growth defects, phenotypes that are only visible under the Nomarski DIC microscope, synthetic phenotypes, etc.) heavily relies on the usage of closely linked, easy-to-score mutations. The traditional method of generating marked mutant alleles is through homologous recombination. Loss-of-function mutations of ced-1, which encodes a C. elegans phagocytic receptor essential for the removal of apoptotic cells (Zhou et al., 2001), result in a cell-corpse persistent phenotype only detectable under the Nomarski DIC microscope. Loss-of-function mutations of unc-75 (Figure 1a) (Loria et al., 2003) cause an Uncoordinated (Unc) phenotype easily detectable under the dissection microscope and are commonly used as linked markers for ced-1. We found that CRISPR/Cas9-mediated knocking out of unc-75 in a ced-1 mutant background is both labor- and time-saving comparing to the traditional method. Here we report our method, result, and the sequence of a small guide RNA (sgRNA) that effectively targets unc-75.We designed three sgRNAs (Figure 1b) predicted to target different regions of unc-75 genomic DNA (Figure 1c) with the help of the online resource (http://crispr.mit.edu/). To express these sgRNA in C. elegans, we cloned each one into plasmid pMB70 (PU6 sgRNA) (Waaijers S et al., 2013). We coinjected all three sgRNA expression constructs (50 ng/µl) together with the Cas9-expression construct pDD162 (50 ng/µl) (Dickinson et al., 2013) and Prab-3 mRFP1 (10 ng/µl) (Mangahas et al., 2008), a transgenic marker, into a ced-1 mutant strain. We found 10 Unc progeny among F1 progeny of 15 injected P0s. Among the progeny of these Unc F1s, we identified three homozygous Unc strains. They did not carry the mRFP1 marker. Sequencing results revealed mutations (Figure 1d) solely in exon 3, within the region targeted by sgRNA3, and no mutations in the regions targeted by sgRNA1 and sgRNA2. Using the same strategy, we isolated additional unc-75 mutant alleles in two other ced-1 mutant backgrounds, in efficiency similar to the above. These alleles also carry mutations solely in the region targeted by sgRNA3 (Figure 1d). In addition, including only sgRNA3 but no sgRNA1 or 2 in the injection mix also resulted in the occurrence, at the similar frequency, of Unc mutations in the F1 generation. The unc-75 mutations we identified include deletions and insertions of up to 16 nucleotides (Figure 1d). Our results thus indicate that sgRNA3 is effective in targeting unc-75 for gene editing, whereas sgRNA1 or sgRNA2 are not.
The frequent occurrence of the Unc-75 mutant worms in the F1 generation indicates that the efficiency of CRISPR/Cas9-mediated gene-editing is high enough to create unc-75 mutations in both homologous alleles. Isolation of Unc mutants in the F1 generation significantly reduces the number of worms to screen through and the time it takes to isolate homozygous mutants. Our observation thus suggests that the CRISPR/Cas9-mediated production of easy-to-score marker mutations is an effective and rapid strategy to generate linked markers that label hard-to-score mutations. We suggest that building a collection of sgRNAs proven successful in targeting commonly used marker genes will benefit the C. elegans community.
Figures
References
Dickinson DJ, Ward JD, Reiner DJ, Goldstein B. (2013). Engineering the Caenorhabditis elegans genome using Cas9-triggered homologous recombination. Nat. Meth. 10, 1028-1034.

Loria PM, Duke A, Rand JB, Hobert O. (2003). Two neuronal, nuclear-localized RNA binding proteins involved in synaptic transmission. Curr. Biol. 13, 1317-1323.

Mangahas PM, Yu X, Miller KG, Zhou Z. (2008). The small GTPase Rab2 functions in the removal of apoptotic cells in Caenorhabditis elegans. J. Cell Biol. 180, 357-373.

Waaijers S, Portegijs V, Kerver J, Lemmens BB, Tijsterman M, et al. (2013). CRISPR/Cas9-targeted mutagenesis in Caenorhabditis elegans. Genetics 195, 1187-1191.

Zhou Z, Hartwieg E, Horvitz HR. (2001). CED-1 is a transmembrane receptor that mediates cell corpse engulfment in C. elegans. Cell 104, 43-56.